
п�̵�أ��׳Ƹɵ�أ��������е������ܴ�����п�̵�صĹ���ͼ��ͼ��a����ʾ��
�ش��������⣺
��1����ͨп�̵�طŵ�ʱ��������Ҫ��ӦΪ��
Zn+2NH4Cl+2MnO2=Zn(NH3)2Cl2+2MnOOH
�ٸõ���У�����������Ҫ��________������ʵ���Ҫ�ɷ���______��������������Ҫ��Ӧ��________��
������ͨп�̵����ȣ�����п�̵�ص��ŵ㼰��������_______��
��2��ͼ��b����ʾ�������÷Ͼ���ͨп�̵�ص�һ�ֹ��գ������ǷϾɵ����ʵ�ʴ��ڵ�����������������
��ͼ��b���в���Ļ�ѧʽ�ֱ�ΪA_______��B________��
�ڲ���a�еõ��ۿ����Ҫ�ɷ���K2MnO4������b�У���ɫ��K2MnO4��Һ��Ӧ��������ɫ��Һ��һ�ֺں�ɫ���壬�÷�Ӧ�����ӷ���ʽΪ_______��
�۲��ö��Ե缫���K2MnO4��ҺҲ�ܵõ�������D�����������õ�����Ҫ������ �����ѧʽ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�������ӷ���ʽ������ȷ����
| A������������ˮ�Ʊ������Cl2��H2O��2H����Cl����ClO�� |
| B����Fe(OH)2�м���������ϡHNO3��Fe(OH)2��2H����Fe2����2H2O |
| C����̼�������Һ�м�������������������Һ��NH4+��OH����NH3��H2O |
| D����������Һ�м������������������Һ��Al3����2SO42����2Ba2����4OH����2BaSO4����AlO2-��2H2O |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��14�֣�����ʯ����Ҫ�ɷ���K2SO4��Al2(SO4)3��2Al2O3��6H2O������������Fe2O3����������ʯ����ȡ������Al2O3��K2FeO4��H2SO4�Ĺ���������ͼ��ʾ��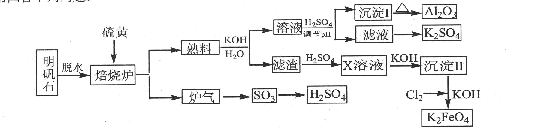
��ش��������⣺
��1������¯�з�����Ӧ�Ļ�ѧ����ʽΪ2Al2(SO4) 2Al2O3+6SO2��+3O2����������102gAl2O3��ת�Ƶĵ�����ĿΪ .
2Al2O3+6SO2��+3O2����������102gAl2O3��ת�Ƶĵ�����ĿΪ .
��2�����ɳ���I�����ӷ���ʽΪ ������II�Ļ�ѧʽΪ ��
��3����Һ�����������pH���ˣ�ϴ�ӣ��ɵó���I��֤������I��ϴ�Ӹɾ���ʵ������������� ��
��4��д��Cl2������������ΪK2FeO4�Ļ�ѧ����ʽ�� ��
��5������Һ�еõ�K2SO4����ķ����� ��
��6�� K2FeO4Ҳ������Ϊ�缫��ͨ�����Ũ��KOH��Һ����ȡ��д����ⷨ��ȡK2FeO4��
������Ӧʽ�� .
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ѧ����ѡ��2����ѧ�뼼������15�֣�
��ˮAlCl3���������������л��ϳɵĴ����ȡ���ҵ����������A12O3��Fe2O3��Ϊԭ���Ʊ���ˮAlCl3�Ĺ����������¡�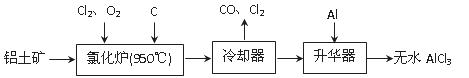
��1���Ȼ�¯��Al2O3��C12��C��Ӧ�Ļ�ѧ����ʽΪ ��
��2����Na2SO3��Һ�ɳ�ȥ��ȴ���ų�β���е�Cl2���˷�Ӧ�����ӷ���ʽΪ ��
��3������������Ҫ����AlCl3��FeCl3�����������Al���������� ��
��4��Ϊ�ⶨ�Ƶõ���ˮAlCl3��Ʒ��������FeCl3���Ĵ��ȣ���ȡ16.25 g��ˮAlCl3��Ʒ�����ڹ�����NaOH��Һ�����˳�����������ᆳϴ�ӡ����ա���ȴ�����أ�������������Ϊ0.32 g��
��д�����ӹ������漰�����ӷ���ʽ �� ��
��AlCl3��Ʒ�Ĵ���Ϊ ��
��5����ҵ����һ��������Ϊԭ���Ʊ���ˮAlCl3�����У����һ������AlCl3��6H2O��ˮ�Ʊ���ˮAlCl3��ʵ����һ���ķ����� ��
��6�������к���һ����̼�����Fe3C������ҵ��Ҫ�ⶨ̼Ԫ�ص�������������һ�����������Ŀ��������գ������д��ԵĹ��壬�÷�Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(1)ij��ɫϡ��ҺX�У����ܺ����±����������е�ij���֡�
| ������ | 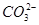 �� ��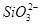 �� ��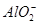 ��Cl�� ��Cl�� |
| ������ | Al3����Cu2����Mg2����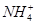 ��Na�� ��Na�� |
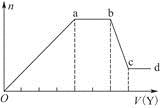
 Sn(OH)2
Sn(OH)2 Sn2����2OH����
Sn2����2OH�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ɫϡ��ҺX�У����ܺ����±����������е�ij���֡�
| ������ | CO32����SiO32����AlO2����Cl�� |
| ������ | Al3����Fe3����Mg2����NH4����Na�� |
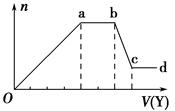
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ʵ������NaHSO4��Ba(OH)2��NH3��H2O��NaHCO3��KAl(SO4)2������
ɫ��Һ������ͨ������֮������Ӧ�����������м��𡣲������ʼ�ķ�Ӧ�������±���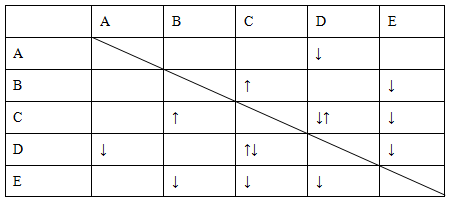
���С�������ʾ�����������ʣ���������ʾ���ɳ�����
����������Ϣ���ش��������⡣
(1)B��E�Ļ�ѧʽ�ֱ�Ϊ________��________��
(2)д��A�ĵ��뷽��ʽ��________��
(3)C��D����Һ��Ӧ�����ӷ���ʽΪ________��
(4)����0.1 mol��Һ��D��Һ�еμ�E��Һ�������ɳ��������ʵ���֮�����Ϊ________mol��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ɫ��Һ����Na����Ag����Ba2����Al3����AlO2����MnO4����CO32����SO42����SiO32���е���
�������(������ˮ�ĵ���)��ȡ����Һ��������ʵ�飺
��.ȡ������Һ��������������ᣬ���������ɣ����õ���ɫ��Һ��
��.�ڢ�������Һ�м��������NH4HCO3��Һ�����������ɣ�ͬʱ������ɫ�����ף�
��.�ڢ�������Һ�м��������Ba(OH)2��Һ�����ȣ�Ҳ���������ɣ�ͬʱ������ɫ�����ҡ�
��ش��������⣺
(1)��ʵ����֪ԭ��Һ��һ�������е�������________��һ�����е�������________________��
(2)��ʵ����֪ԭ��Һ�л�һ�����е�������________�����ɼ����ӷ���ʽΪ________________��
(3)ʵ��������ɰ�ɫ�����ҵ����ӷ���ʽΪ________________��
(4)ԭ��Һ�л����ܴ��ڵ�������________����������ӵķ�����_________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ú���Al2O3��SiO2������FeO��xFe2O3�������Ʊ�Al2(SO4)3��18H2O��������������(���ֲ�����������)��
��.�������м������ϡH2SO4�����ˣ�
��.����Һ�м������KMnO4��Һ��������Һ��pHԼΪ3��
��.���ȣ�����������ɫ���������ã��ϲ���Һ���Ϻ�ɫ��
��������MnSO4���Ϻ�ɫ��ʧ�����ˣ�
V��Ũ�����ᾧ�����룬�õ���Ʒ��
(1)H2SO4�ܽ�Al2O3�����ӷ���ʽ��_______________________��
(2)��MnO4������Fe2�������ӷ���ʽ����������
1MnO4����Fe2����________===1Mn2����Fe3����________
(3)��֪��
�����������������pH
| | Al(OH)3 | Fe(OH)2 | Fe(OH)3 |
| ��ʼ����ʱ | 3.4 | 6.3 | 1.5 |
| ��ȫ����ʱ | 4.7 | 8.3 | 2.8 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com