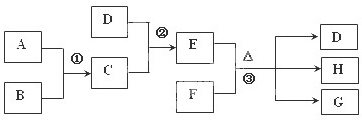
�⣺A���峣���³ʻ���ɫ����AΪCl
2��A��B��ȼ�յĻ���ʲ�ɫ��BΪH
2��CΪHCl��D�����ˮ��Һ�ʼ��ԣ���DΪNH
3������EΪNH
4Cl��F��Һ�׳�ʯ��ˮ����FΪCa��OH��
2��GΪ�Ȼ��ƣ���HΪˮ��
��1��������������֪��AΪCl
2��BΪH
2��DΪNH
3��ͬ��10���ӵ������ӻ�ѧʽ�ֱ�ΪNH
4+��H
3O
+���ʴ�Ϊ��Cl
2��H
2��NH
3��NH
4+��H
3O
+��
��2��D����ˮ��Һ�ĵ��뷽��ʽΪNH
3��H
2O?NH
4++OH
-��E��ˮ�ⷴӦ���ӷ���ʽΪNH
4++H
2O?NH
3��H
2O+H
+��
�ʴ�Ϊ��NH
3��H
2O?NH
4++OH
-��NH
4++H
2O?NH
3��H
2O+H
+��
��3����Ӧ��ΪCl
2+H
2�T2HCl����Ӧ��ΪHCl+NH
3�TNH
4Cl����Ӧ��ΪCa��OH��
2+2NH
4Cl

CaCl
2+2NH
3��+2H
2O��ֻ�з�Ӧ������Ԫ�ػ��ϼ۱仯������������ԭ��Ӧ��
�ʴ�Ϊ���٣�
��4����Ӧ��ΪHCl+NH
3�TNH
4Cl����Ӧ��ΪCa��OH��
2+2NH
4Cl

CaCl
2+2NH
3��+2H
2O���ʴ�Ϊ��HCl+NH
3�TNH
4Cl��Ca��OH��
2+2NH
4Cl

CaCl
2+2NH
3��+2H
2O��
������A���峣���³ʻ���ɫ����AΪCl
2��A��B��ȼ�յĻ���ʲ�ɫ��BΪH
2��CΪHCl��D�����ˮ��Һ�ʼ��ԣ���DΪNH
3������EΪNH
4Cl��F��Һ�׳�ʯ��ˮ����FΪCa��OH��
2��GΪ�Ȼ��ƣ���HΪˮ��Ȼ����Ԫ�ػ��������ʼ���ѧ���������
���������⿼��������ƶϣ�ע��������Ϣ�ƶϸ������ǽ��Ĺؼ�����ȷ���������ʼ��������Ʒ����ɽ����ɫΪ����ͻ�ƿڣ���Ŀ�Ѷ��еȣ�

 CaCl2+2NH3��+2H2O��ֻ�з�Ӧ������Ԫ�ػ��ϼ۱仯������������ԭ��Ӧ��
CaCl2+2NH3��+2H2O��ֻ�з�Ӧ������Ԫ�ػ��ϼ۱仯������������ԭ��Ӧ�� CaCl2+2NH3��+2H2O���ʴ�Ϊ��HCl+NH3�TNH4Cl��Ca��OH��2+2NH4Cl
CaCl2+2NH3��+2H2O���ʴ�Ϊ��HCl+NH3�TNH4Cl��Ca��OH��2+2NH4Cl CaCl2+2NH3��+2H2O��
CaCl2+2NH3��+2H2O��

 Сѧ�̲���ȫ���ϵ�д�
Сѧ�̲���ȫ���ϵ�д�









 ��֪A��B��C��D��Ϊ���壬E��F��Ϊ�����³ʹ�������ӻ����GΪ�Ȼ��ƣ�A��B��ȼ�յĻ���ʲ�ɫ����Ӧ���������������д������̣�����֮���ת����ϵ��ͼ��ʾ��
��֪A��B��C��D��Ϊ���壬E��F��Ϊ�����³ʹ�������ӻ����GΪ�Ȼ��ƣ�A��B��ȼ�յĻ���ʲ�ɫ����Ӧ���������������д������̣�����֮���ת����ϵ��ͼ��ʾ��





