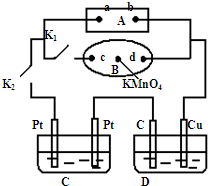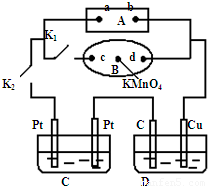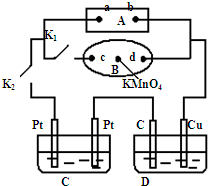
��1����ͼ��ʾ��AΪ��Դ��BΪ������ʳ��ˮ�ͷ�̪��Һ����ֽ����ֽ�������һ��KMnO
4��Һ��C��DΪ���ۣ���缫���ϼ�ͼ���������Һ��Cװ������ҺΪAgNO
3��Һ��Dװ�õ���ҺΪCu��NO
3��
2��Һ���ر�K
1����K
2��ͨ���B��KMnO
4�Ϻ�ɫҺ����d���ƶ������Դa��Ϊ
��
��
����ͨ��һ��ʱ��۲쵽��ֽc�˳��ֵ������ǣ�
���ɫ
���ɫ
��
��2����K
1���ر�K
2��ͨ��һ��ʱ���Dװ����Cu���ĵ缫��ӦʽΪ
Cu-2e-=Cu2+
Cu-2e-=Cu2+
��
Cװ�����Ҷ�Pt�缫�ϵĵ缫��ӦʽΪ��
4OH--4e-=2H2O+O2��
4OH--4e-=2H2O+O2��
��
Cװ�����ܷ�Ӧ����ʽΪ��
4AgNO
3+2H
2O
4Ag+O
2��+4HNO
3 4AgNO
3+2H
2O
4Ag+O
2��+4HNO
3 ��
��3���ٶ�C��Dװ���е������Һ���������һ��ʱ���Cװ������Һ��pHֵ
��С
��С
����������С�����䡱����ͬ����Dװ������Һ��pHֵ
����
����
��

 ��1����ͼ��ʾ��AΪ��Դ��BΪ������ʳ��ˮ�ͷ�̪��Һ����ֽ����ֽ�������һ��KMnO4��Һ��C��DΪ���ۣ���缫���ϼ�ͼ���������Һ��Cװ������ҺΪAgNO3��Һ��Dװ�õ���ҺΪCu��NO3��2��Һ���ر�K1����K2��ͨ���B��KMnO4�Ϻ�ɫҺ����d���ƶ������Դa��Ϊ
��1����ͼ��ʾ��AΪ��Դ��BΪ������ʳ��ˮ�ͷ�̪��Һ����ֽ����ֽ�������һ��KMnO4��Һ��C��DΪ���ۣ���缫���ϼ�ͼ���������Һ��Cװ������ҺΪAgNO3��Һ��Dװ�õ���ҺΪCu��NO3��2��Һ���ر�K1����K2��ͨ���B��KMnO4�Ϻ�ɫҺ����d���ƶ������Դa��Ϊ


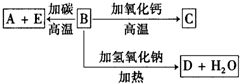 A��B��C��D��E�������ʻ������֮����ת����ϵ��ͼ��ʾ��AΪ�ؿ��к������������ķǽ���Ԫ�صĵ��ʣ��侧��ṹ����ʯ���ƣ�
A��B��C��D��E�������ʻ������֮����ת����ϵ��ͼ��ʾ��AΪ�ؿ��к������������ķǽ���Ԫ�صĵ��ʣ��侧��ṹ����ʯ���ƣ�

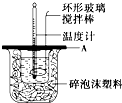 �ֱ�ȡ40mL��0.50mol/L������40mL��0.55mol/L����������Һ�����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ���ش��������⣮
�ֱ�ȡ40mL��0.50mol/L������40mL��0.55mol/L����������Һ�����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ���ش��������⣮