��2012?���X��Y��Z��M��G����Ԫ�ط������������ڣ���ԭ��������������X��Zͬ���壬���γ����ӻ�����ZX��Y��Mͬ���壬���γ�MY
2��MY
3���ַ��ӣ�
��ش��������⣺
��1��Y��Ԫ�����ڱ��е�λ��Ϊ
�ڶ����ڵڢ�A��
�ڶ����ڵڢ�A��
��
��2������Ԫ�ص�����������Ӧ��ˮ����������ǿ����
HClO4
HClO4
��д��ѧʽ�����ǽ�����̬�⻯�ﻹԭ����ǿ����
H2S
H2S
��д��ѧʽ����
��3��Y��G�ĵ��ʻ���Ԫ��֮���γɵĻ��������ˮ����������
O3��Cl2��
O3��Cl2��
��д�������������ʵĻ�ѧʽ����
��4��X
2M��ȼ���ȡ�H=-a kJ?mol
-1���X
2Mȼ�շ�Ӧ���Ȼ�ѧ����ʽ��
2H2S��g��+3O2��g��=2SO2��g��+2H2O��l������H=-2aKJ?mol-1
2H2S��g��+3O2��g��=2SO2��g��+2H2O��l������H=-2aKJ?mol-1
��
��5��ZX�ĵ���ʽΪ
��ZX��ˮ��Ӧ�ų�����Ļ�ѧ����ʽΪ
NaH+H2O=NaOH+H2��
NaH+H2O=NaOH+H2��
��
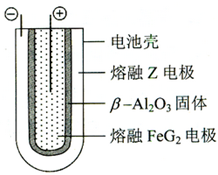
��6������״̬�£�Z�ĵ��ʺ�FeG
2����ɿɳ���أ�װ��ʾ��ͼ���£�����Ӧԭ��Ϊ��2Z+FeG2
Fe+2ZG�� �ŵ�ʱ����ص�������ӦʽΪ
Fe2++2e-=Fe
Fe2++2e-=Fe
�����ʱ��
Na
Na
��д�������ƣ��缫�ӵ�Դ�ĸ������õ�صĵ����Ϊ
��-Al2O3
��-Al2O3
��

 ��2012?���X��Y��Z��M��G����Ԫ�ط������������ڣ���ԭ��������������X��Zͬ���壬���γ����ӻ�����ZX��Y��Mͬ���壬���γ�MY2��MY3���ַ��ӣ�
��2012?���X��Y��Z��M��G����Ԫ�ط������������ڣ���ԭ��������������X��Zͬ���壬���γ����ӻ�����ZX��Y��Mͬ���壬���γ�MY2��MY3���ַ��ӣ�

 ��Na��ˮ��Ӧ��������������������Ӧ��ѧ����ʽΪΪ��NaH+H2O=NaOH+H2����
��Na��ˮ��Ӧ��������������������Ӧ��ѧ����ʽΪΪ��NaH+H2O=NaOH+H2���� ��NaH+H2O=NaOH+H2����
��NaH+H2O=NaOH+H2����


 ��2012?���ģ�⣩������Ԫ��X��Y��Z��W��Q��Ԫ�����ڱ��е����λ����ͼ��ʾ������˵����ȷ���ǣ�������
��2012?���ģ�⣩������Ԫ��X��Y��Z��W��Q��Ԫ�����ڱ��е����λ����ͼ��ʾ������˵����ȷ���ǣ�������




