
 =7.14 mol��L��1��
=7.14 mol��L��1��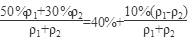 ��40%��
��40%��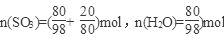 ��
�� =0.04mol����ͨ��112mL(��״��)����ǡ�ý�Fe2����ȫ����(2Fe2��+Cl2==2Fe3++2Cl-)��n(Fe2+)=2��
=0.04mol����ͨ��112mL(��״��)����ǡ�ý�Fe2����ȫ����(2Fe2��+Cl2==2Fe3++2Cl-)��n(Fe2+)=2��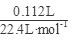 =0.01mol���ɵ���غ�n(Fe3+)=0.02mol�� 7.32�˾��庬��0.01molFeSO4Ϊ1.52g��0.01mol Fe2(SO4)3Ϊ4.00g������H2OΪ1.80g��0.1mol������Ļ�ѧʽFeSO4��Fe2(SO4)3��10H2O��
=0.01mol���ɵ���غ�n(Fe3+)=0.02mol�� 7.32�˾��庬��0.01molFeSO4Ϊ1.52g��0.01mol Fe2(SO4)3Ϊ4.00g������H2OΪ1.80g��0.1mol������Ļ�ѧʽFeSO4��Fe2(SO4)3��10H2O��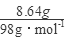 =0.09mol����Ҫ��������ʵ���Ϊy��
=0.09mol����Ҫ��������ʵ���Ϊy�� ��100%��33.33%��
��100%��33.33%��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��FeCl2 | B��CuCl2 | C��HCl | D��H2O |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����FeSO4��Һ�м���NaOH��Һ����������Ƶ�Fe��OH��2 |
| B��Al2O3�ܺ�NaOH��Һ��Ӧ���õ�NaAlO2 |
| C��ij��Һ��ɫ��Ӧ�Ļ���ʻ�ɫ�������Һ�в��������� |
| D��ij��Һ�м���BaCl2��Һ�а�ɫ�����������ټ����ᣬ��������ʧ�������Һ��һ����SO42- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
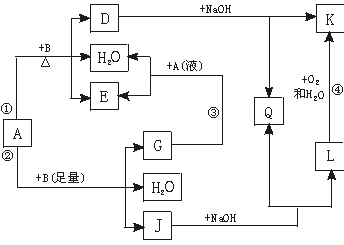
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ƶ���
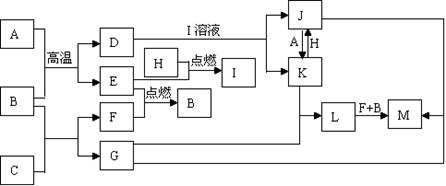
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
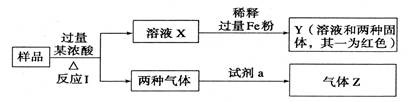
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
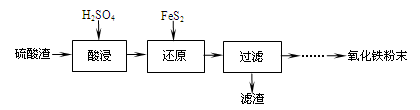
| ������ | Fe(OH)3 | Al(OH)3 | Fe(OH)2 | Mg(OH)2 | Mn(OH)2 |
| ��ʼ���� | 2.7 | 3.8 | 7.5 | 9.4 | 8.3 |
| ��ȫ���� | 3.2 | 5.2 | 9.7 | 12.4 | 9.8 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ȡ������������茶�������Թ��У����ȣ��Թܿ���Һ�����ɣ����֤������ijɷ��к��нᾧˮ |
| B��ȡ������������茶�������Թܣ���ŨNaOH��Һ�����ȣ��Թܿ�ʪ�����ɫʯ����ֽ��죬���֤������ijɷ��к���NH4+ |
| C��ȡ������������茶�������ˮ��������ϡ���ᣬ�������ٵ��뼸��BaCl2��Һ���а�ɫ�������ɣ����֤������ijɷ��к���SO42- |
| D��ȡ������������茶�������ˮ����dz��ɫ��Һ������2��KSCN��Һ����Һ����Ѫ��ɫ���ٵ��뼸��������ˮ����Һ��ΪѪ��ɫ�����֤������ijɷ��к���Fe2�� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com