
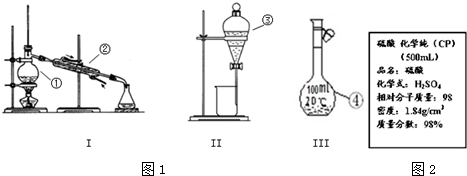
·ÖĪö £Ø1£©øł¾ŻÅäÖĘ100mL 0.9mol/LµÄĻ”ĮņĖįµÄ²½Öč¶Ōø÷²Ł×÷½ųŠŠÅÅŠņ£»
£Ø2£©øł¾Żc=$\frac{1000¦Ńw}{M}$¼ĘĖćÅØĮņĖįµÄĪļÖŹµÄĮæÅØ¶Č£¬ŌŁøł¾ŻĻ”ŹĶ¶ØĀɼĘĖćŠčŅŖÅØĮņĖįµÄĢå»ż£»
£Ø3£©øł¾Żc=$\frac{n}{V}$æÉµĆ£¬Ņ»¶ØĪļÖŹµÄĮæÅضČČÜŅŗÅäÖʵÄĪó²ī¶¼ŹĒÓÉČÜÖŹµÄĪļÖŹµÄĮænŗĶČÜŅŗµÄĢå»żVŅżĘšµÄ£¬Īó²ī·ÖĪöŹ±£¬¹Ų¼üŅŖæ“ÅäÖĘ¹ż³ĢÖŠŅżĘšnŗĶVŌõŃłµÄ±ä»Æ£ŗČōn±ČĄķĀŪÖµŠ”£¬»ņV±ČĄķĀŪÖµ“óŹ±£¬¶¼»įŹ¹ĖłÅäČÜŅŗÅضČĘ«Š”£»Čōn±ČĄķĀŪÖµ“󣬻ņV±ČĄķĀŪÖµŠ”Ź±£¬¶¼»įŹ¹ĖłÅäČÜŅŗÅضČĘ«“ó£®
½ā“š ½ā£ŗ£Ø1£©ÅäÖĘ100mL 0.9mol/LµÄĻ”ĮņĖįµÄ²½ÖčĪŖ£ŗ¼ĘĖć”¢ĮæČ””¢Ļ”ŹĶ”¢ĄäČ“”¢ŅĘŅŗ”¢Ļ“µÓ”¢¶ØČŻŗĶŅ”ŌČµČ£¬ÕżČ·µÄ²Ł×÷Ė³ŠņĪŖ£ŗADECBHGF£¬
¹Ź“š°øĪŖ£ŗADECBHGF£»
£Ø2£©£©98%µÄÅØĮņĖį£ØĆܶČĪŖ1.8g•cm-3£©µÄĪļÖŹµÄĮæÅضČC=$\frac{1000”Į1.8g/ml”Į98%}{98g/mol}$=18mol/L£¬ÉčŠčŅŖÅØĮņĖįĢå»żĪŖV£¬ŅĄ¾ŻČÜŅŗĻ”ŹĶ¹ęĀÉæÉµĆ£ŗV”Į18mol/L=3.6mol•L-1”Į250ml£¬½āµĆ£ŗV=50.0ml£»
¹Ź“š°øĪŖ£ŗ50.0£»
£Ø3£©A£®Ź¹ÓĆČŻĮæĘæÅäÖĘČÜŅŗŹ±£¬ø©ŹÓŅŗĆę£¬¼ÓČėµÄÕōĮóĖ®µĶÓŚČŻĮæĘææĢ¶ČĻߣ¬µ¼ÖĀÅäÖʵÄČÜŅŗĢå»żĘ«Š”£¬ĖłµĆČÜŅŗµÄÅضČĘ«“󣬹ŹAÕżČ·£»
B£®Ć»ÓŠÓĆÕōĮóĖ®Ļ“ÉÕ±2-3“Ī£¬²¢½«Ļ“ŅŗŅĘČėČŻĮæĘæÖŠ£¬»įµ¼ÖĀÅäÖʵÄČÜŅŗÖŠČÜÖŹµÄĪļÖŹµÄĮæĘ«Š”£¬ÅäÖʵÄČÜŅŗÅضČĘ«µĶ£¬¹ŹB“ķĪó£»
C£®ČŻĮæĘæÓĆÕōĮóĖ®Ļ“¾»£¬Ć»ÓŠŗęøÉ£¬ČŻĮæĘæ²»ŠčŅŖŗęøÉ£¬¶ŌČÜÖŹµÄĪļÖŹµÄĮæ¼°ČÜŅŗµÄ×īÖÕĢå»ż²»±ä£¬²»Ó°ĻģÅäÖĘ½į¹ū£¬¹ŹC“ķĪó£»
D£®¶ØČŻŹ±£¬µĪ¼ÓÕōĮóĖ®£¬ĻČŹ¹ŅŗĆęĀŌøßÓŚæĢ¶ČĻߣ¬µ¼ÖĀÅäÖʵÄČÜŅŗĢå»żĘ«“ó£¬ÅäÖʵÄČÜŅŗÅضČĘ«µĶ£¬¹ŹD“ķĪó£»
E£®°ŃÅäŗƵÄČÜŅŗµ¹ČėÓĆÕōĮóĖ®Ļ“¾»¶ųÄ©øɵďŌ¼ĮĘæÖŠ±øÓĆ£¬ŹŌ¼ĮĘæÖŠµÄÕōĮóĖ®½«ÅäÖʵÄČÜŅŗĻ”ŹĶ£¬µ¼ÖĀČÜŅŗÅضČĘ«Š”£¬¹ŹE“ķĪó£»
¹Ź“š°øĪŖ£ŗA£®
µćĘĄ ±¾Ģāæ¼²éĮĖÅäÖĘŅ»¶ØĪļÖŹµÄĮæÅØ¶ČµÄČÜŅŗµÄ·½·Ø£¬ÄѶČÖŠµČ£¬ŹŌĢāÄŃŅ׏ŹÖŠ£¬×¢ÖŲĮé»īŠŌ£¬²ąÖŲ¶ŌѧɜÄÜĮ¦µÄÅąŃųŗĶ½āĢā·½·ØµÄÖøµ¼ŗĶѵĮ·£¬ÓŠĄūÓŚÅąŃųѧɜµÄĀß¼Ė¼Ī¬ÄÜĮ¦ŗĶŃĻ½÷µÄ¹ę·¶ŹµŃé²Ł×÷ÄÜĮ¦£¬øĆĢāµÄÄѵćŌŚÓŚĪó²ī·ÖĪö£¬×¢ŅāĆ÷Č·Īó²ī·ÖĪöµÄ·½·Ø£®



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
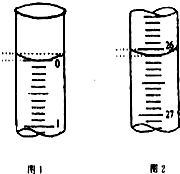 Ä³Ń§ÉśÓūÓĆŅŃÖŖĪļÖŹµÄĮæÅØ¶ČµÄŃĪĖįĄ“²ā¶ØĪ“ÖŖĪļÖŹµÄĮæÅØ¶ČµÄĒāŃõ»ÆÄĘČÜŅŗŹ±£¬Ń”Ōń¼×»ł³Č×÷ÖøŹ¾¼Į£®ĒėĢīŠ“ĻĀĮŠæÕ°×£ŗ
Ä³Ń§ÉśÓūÓĆŅŃÖŖĪļÖŹµÄĮæÅØ¶ČµÄŃĪĖįĄ“²ā¶ØĪ“ÖŖĪļÖŹµÄĮæÅØ¶ČµÄĒāŃõ»ÆÄĘČÜŅŗŹ±£¬Ń”Ōń¼×»ł³Č×÷ÖøŹ¾¼Į£®ĒėĢīŠ“ĻĀĮŠæÕ°×£ŗ| µĪ¶Ø“ĪŹż | “ż²āĒāŃõ»ÆÄĘČÜŅŗµÄĢå»ż/mL | 0.1000mol•L-1ŃĪĖįµÄĢå»ż/mL | ||
| µĪ¶ØĒ°æĢ¶Č | µĪ¶ØŗóæĢ¶Č | ČÜŅŗĢå»ż/mL | ||
| µŚŅ»“Ī | 25.00 | 0.00 | 26.11 | 26.11 |
| µŚ¶ž“Ī | 25.00 | 1.56 | 30.30 | 28.74 |
| µŚČż“Ī | 25.00 | 0.22 | 26.31 | 26.09 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
| Cu£ØOH£©2 | CuOH | CuCl | Cu2O | |
| ŃÕÉ« | Ą¶É« | »ĘÉ« | °×É« | שŗģÉ« |
| Ksp£Ø25”ę£© | 1.6”Į10-19 | 1.0”Į10-14 | 1.2”Į10-6 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
| “ĪŹż | ²āĮæĻīÄæ | ²āĮæŹż¾Ż£Ømg/mL£© |
| 1 | ĶĄė×Óŗ¬Įæ | 4.224 |
| 2 | 4.225 | |
| 3 | 4.223 | |
| 4 | 2.005 |

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā

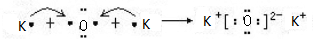
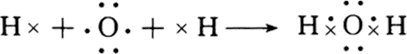
 £®
£®²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | Cl2ÓėH2O·“Ó¦£ŗCl2+H2OØT2H++ClO-+Cl- | |
| B£® | ĶłFeCl3ČÜŅŗÖŠ¼ÓČėCu·Ū£ŗFe3++CuØTFe2++Cu2+ | |
| C£® | ĻņAlCl3ČÜŅŗÖŠµĪ¼Ó°±Ė®ÖĮ¹żĮæ£ŗAl3++3OH-ØTAl£ØOH£©3”ż | |
| D£® | ½«ĶʬĶ¶ČėÅØĻõĖįÖŠ£ŗ2NO3-+Cu+4H+ØTCu2++2NO2”ü+2H2O |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com