
��14�֣�����ΪԪ�����ڱ��е�һ���֣��û�ѧʽ��Ԫ�ط��Żش��������⣺
| �� ���ڡ��� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| �� |
|
|
| �� |
| �� |
| |
| �� | �� | �� | �� |
|
|
| �� | �� |
| �� | �� | �� |
|
|
|
| �� |
|
(1) �������Ԫ���γɵ���̬�⻯�����ȶ�����___________________________��
(2) д���ٵ�����������Ӧ��ˮ����ĵ���ʽ________ ��
(3) �ڢۢ����γɵļ����Ӱ뾶�ɴ�С��˳����________________________��
(4) �õ���ʽ��ʾԪ�آߵ��⻯����γɹ���________ �����⻯����Ԫ�آ�ĵ��ʷ�Ӧ�����ӷ���ʽ��______________________________��
(5) �ٺ͢���Ԫ���γɻ�����Ļ�ѧʽΪ_________________________���û��������Һ��Ԫ�آ�ĵ��ʷ�Ӧ�����ӷ���ʽΪ___________________ ��
��1��HF ��2�� ��3��K����Ca2����Mg2��
��4����Cl2��H2O=HCl��HclO
��5��NaBr 2Br����Cl2=Br2��2Cl��
����:����Ԫ�����ڱ��Ľṹ��Ԫ�������ɵ�Ӧ�á�����Ԫ�������ڱ��е�λ�ÿ�֪����Na������K������Mg������Ca������Al������C������O������Cl������Br������Ar����F��
��1��ͬ����Ԫ�����϶��·ǽ������������⻯����ȶ���������
��2���Ƶ�����������Ӧ��ˮ�������������ƣ������ӻ����
��3��ͬ����Ԫ�����϶���ԭ�Ӱ뾶�ͣ����Ӱ뾶����������ġ�ͬ����Ԫ����������ԭ�Ӱ뾶��С�������Ӱ뾶��Ҳ��С������˳��ΪK����Ca2����Mg2����
��4��O���⻯����ˮ���γɵĻ�ѧ���Ǽ��Լ����γɹ���Ϊ
����������ˮ�����Ȼ���ʹ����ᣬ����ʽΪCl2��H2O=HCl��HClO��
��5���ȵķǽ�����ǿ����ģ������Խ��������������ɵ��ʵ����壬��ӦʽΪ2Br����Cl2=Br2��2Cl����


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ��A | 0 | |||||||
| 1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� | |||||||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ��A | 0 | |||||||
| 1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | |
| 2 | �� | �� | �� | �� | ||||
| 3 | �� | �� | �� | �� | ||||
| 4 | =10 �� | |||||||
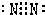
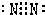
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �� ���� |
IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 |
| �� | �� | �� | �� | �� | ||||
| �� | �� | �� | �� | �� | �� | |||
| �� | �� |  |
 ��Ԫ�غ˵��֮����
��Ԫ�غ˵��֮�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ������ | ��A | ��A | ��A | ��A | ��A | ��A | ��A |
0 |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | ||||
| 4 | �� | �� |


�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com