




| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
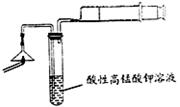
| A���ڢ��У�����������������Һ�ͳ����� |
| B���ڢ��У����PH��7������������ɷ���NaFˮ������ģ� |
| C���ڢ��У�������Cu(OH)2�����������ɫ������ |
| D���ڢ��У��ų�������ͨ������ʯ��ˮ��һ�����ֳ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
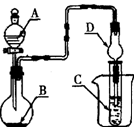
| A����AΪŨ���ᣬBΪMnO2��C��ʢƷ����Һ����C����Һ��ɫ |
| B����AΪϡ���ᣬBΪʯ��ʯ��C��ʢCaCl2��Һ����C����Һ����� |
| C����AΪŨ��ˮ��BΪ��ʯ�ң�C��ʢA1C13��Һ����C���Ȳ�����ɫ������������ܽ� |
| D����AΪŨ���ᣬBΪͭм��C��ʢ��ɫʯ����Һ����C����Һ���ɫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
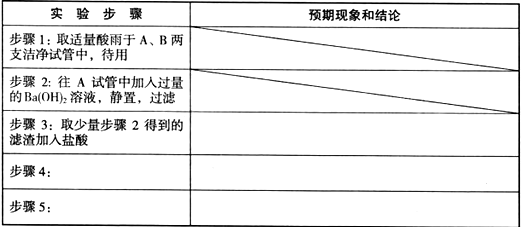
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
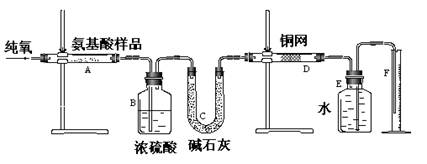
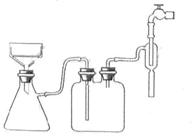
| A�����ɶ�����̼��������� | B������ˮ������ |
| C��ͨ����������� | D�����������Է������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| V/(NaOH)/mL | 0.00 | 10.00 | 18.00 | 19.80 | 19.98 | 20.00 | 20.02 | 20.20 | 22.00 | 40.00 |
| ��ҺpH | 2.87 | 4.74 | 5.70 | 6.74 | 7.74 | 8.72 | 9.70 | 10.70 | 11.70 | 12.50 |
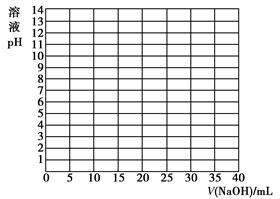
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 ��죬�ּ�����ʹ�����ǵ�ľ����ȼ������0.28L������£���
��죬�ּ�����ʹ�����ǵ�ľ����ȼ������0.28L������£����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
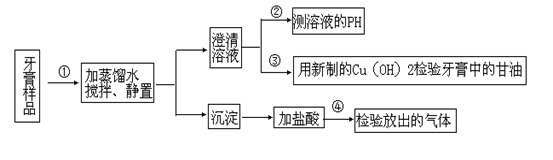
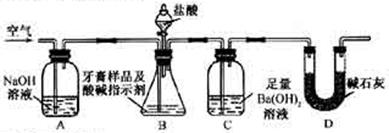
 ��ijƷ��������Ħ�����ɷּ��京����������̽����
��ijƷ��������Ħ�����ɷּ��京����������̽���� __________________________________________��
__________________________________________��
 ___��
___�� ��Ƶ���������Ϊ__________��
��Ƶ���������Ϊ__________���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| �ζ����� | ������Һ�����/mL | ����Һ�����/mL | |
| �ζ�ǰ�̶� | �ζ���̶� | ||
| 1 | 25.00 | 1.02 | 21.03 |
| 2 | 25.00 | 2.00 | 21.99 |
| 3 | 25.00 | 0.20 | 20.20 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com