
�Խ�����Ʒ���п���ʴ���������ӳ���ʹ��������
��1������Ϊ���ı��洦����һ�ַ�����
�ټ�ϴ��Ŀ����ϴȥ���ı������Ȼ����Ĥ����ϴʱ��������ð����ԭ�������� �������� �������ӷ���ʽ��ʾ����
Ϊ����ϴ��Һ�е����Գ�����ʽ���գ�������Һ�м��������Լ��е��������� ��
a��NH3�������� b��CO2 ����������c�� NaOH������ ����d��HNO3
��������Ϊ�������ڹ�����H2SO4��Һ�е�⣬���ı����γ�����Ĥ�������缫��ӦʽΪ������ ��ȡ�����ϵ��Һ������NaHCO3��Һ��������ݺͰ�ɫ������ԭ���� ������������ �� �������������� �������ӷ���ʽ��ʾ����
��2����ͭ�ɷ�ֹ����Ʒ��ʴ�����ʱ��ͭ������ʯī��������ԭ����
��3�����������װ�ã�����ģ�����ĵ绯ѧ��������XΪ̼����Ϊ�������ĸ�ʴ������KӦ�������� ������
��XΪп������K����M�����õ绯ѧ��������Ϊ������������������������������ ��
��1����2Al+2OH��+2H2O��2AlO2��+3H2����1�֣� b(1��)
��2Al-6e��+3H2O��Al2O3��6H����2�֣�
HCO3���� H���� CO2��+ H2O ��2�֣�
Al3++3HCO3����Al(OH)3��+3CO2����2�֣�
��2��������Һ�����ĵ�Cu2+��������Һ��Cu2+Ũ�Ⱥ㶨����2�֣�
��3��N��1�֣���������������������������������������������1�֣�
����������������ļ��Կ���ϴȥ���ۣ�ͬʱҲ����ϴȥ���������������¶������ͼ���Һ��Ӧ�ų�������2Al+2OH��+2H2O��2AlO2��+3H2�������������������������������ǿ��ǿ���ˣ���Һ�е�ƫ�������Ӧͨ��CO2ʹ�����������������ѡb����������Ϊ�������ڹ�����H2SO4��Һ�е�⣬���ı����γ�����Ĥ����ʧ���ӷ���������Ӧ�������缫��ӦʽΪ2Al-6e��+3H2O��Al2O3��6H����ȡ�����ϵ��Һ������NaHCO3��Һ��������ݺͰ�ɫ������ԭ����Һ�������ᣬ������������HCO3���� H���� CO2��+ H2O��Al3++3HCO3����Al(OH)3��+3CO2������2����ͭ�ɷ�ֹ����Ʒ��ʴ�����ʱ��ͭ������ʯī��������ԭ���Dz�����Һ�����ĵ�Cu2+��������Һ��Cu2+Ũ�Ⱥ㶨����3����XΪ̼����Ϊ�������ĸ�ʴ��Ӧ���ɵ��أ������ĵ籣����������KӦ������N������XΪп������K����M�����õ绯ѧ��������Ϊ����������������������
���㣺���鿼����������仯��������ʡ�ԭ���ԭ���ͽ����ķ���


 ��ս100��Ԫ����Ծ�ϵ�д�
��ս100��Ԫ����Ծ�ϵ�д� ������ϵ�д�
������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�Ϻ�ijС���ϣ���ž�սʿΪ��Ѱ�Һ��ʵ�����ˮԴ���Ե���ɽȪˮ���з������飬�����ʾˮ��Ӳ��Ϊ28(����Ӳˮ)����Ҫ�������ӡ�þ���ӡ������Ӻ���������ӡ���˼���������⣺
��1����Ȫˮ����_________Ӳˮ����д����ʱ�������á�����
��2����Ҫ��ȥCa2����Mg2��������ˮ�м���ʯ�Һʹ���Լ�����ʱ�ȼ�________ ���________��ԭ����_____________________________________________________________��
��3��Ŀǰ���������ӽ�����֬��NaR��HR������ˮ����������ʹ��HR��Ϊ�����ӽ�����֬�� ��ˮ�е�Ca2����Mg2���뽻����֬��________�����ӽ������á���ʹ��NaR��Ϊ�����ӽ�����֬��ʧЧ��ɷ���5%��8%_____________��Һ��������
��4�����ϻ������ú�ˮ��������õ�ˮ�������Ǻ�ˮ���õ���������õ�ˮ��ԭ��ͼ����֪��ˮ�к�Na����Cl����Ca2����Mg2����SO42�������ӣ��缫Ϊ���Ե缫��������������⣺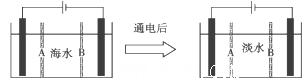
�������ӽ���Ĥ��ָ______________(��A��B)��
��д��ͨ����������ĵ缫��Ӧʽ��_____________________________.
�������������ǣ�_______________________________________________.
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��.��1�����ʱ���Ƽ����Դ��________�����ӡ�
��2����ѧ�Ƶ�ԭ�������û�ѧ��Ӧ���ɽ������ʳ����ڶƼ������γɶƲ㡣����ͭ�ν��л�ѧ��ͭ��Ӧѡ��________(�����������ԭ����)��֮��Ӧ��
��.����[CO(NH2)2]�����������˹��ϳɵ��л���˹�����ɲ��ü�ӵ绯ѧ������ȥ��л�����е����أ�ԭ����ͼ��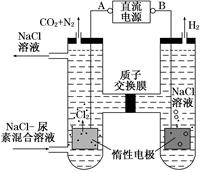
��1����Դ�ĸ���Ϊ________(�A����B��)��
��2���������з����ķ�Ӧ����Ϊ________��________��
��3������������������Һ��pH����ǰ��Ƚ�________�����������ռ�������13.44 L(��״��)�����ȥ������Ϊ________g(����������ܽ�)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ��ʾ��ijͬѧ�����һ��ȼ�ϵ�ز�̽���ȼҵ��ԭ���ʹ�ͭ�ľ���ԭ����������װ����XΪ�����ӽ���Ĥ���밴Ҫ��ش�������⣺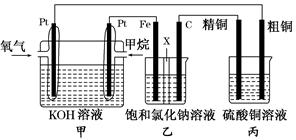
(1)����ȼ�ϵ�ظ�����ӦʽΪ________________________��
(2)ʯī(C)���ĵ缫��ӦʽΪ_______________________��
(3)���ڱ�״���£���2.24 L�����μӷ�Ӧ������װ�������������ɵ���������Ϊ________L����װ������������ͭ������Ϊ________ g��
(4)ijͬѧ���ü���ȼ�ϵ����Ƶ�ⷨ��ȡƯ��Һ��Fe(OH)2��ʵ��װ��(��ͼ��ʾ)��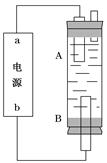
��������Ư��Һ��aΪ��ص�________�����������Һ�����)________________
��������Fe(OH)2��ʹ����������Һ���������Һ������ѡ��________���缫��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������ǿ������������ԭ�ֶΣ��ڻ���������������Ҫ��Ӧ�á���ش��������⣺
��1����ͭΪ��������ʯīΪ��������NaCl��Һ�����Һ���е�⣬�õ��뵼�����Cu2O��һ�������Դ����������ӦʽΪ___________________________��������ӦʽΪ________��
��2��ijͬѧ�����ͼ��ʾ��װ��̽�������ĸ�ʴ����������жϺ�������____________������ţ���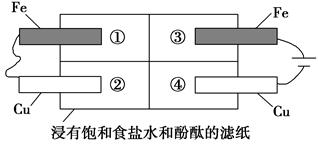
a������ͭƬ�������ݲ���
b��������Ƭ�ĵ缫��ӦʽΪ2Cl����2e��=Cl2��
c�����ȹ۲쵽��ɺ�ɫ�������Ǣ���
d�������͢�����ͭƬ���������������仯
��3�������о����֣��ø�Ĥ��ⷨ������Ũ����ȩ��ˮ�Ĺ��վ������̼��ܺĽϵ͵��ŵ㣬��ԭ����ʹ��ȩ�ֱ�����������������Ӧ�����Ҵ������ᣬ�ܷ�ӦʽΪ2CH3CHO��H2O CH3CH2OH��CH3COOH
CH3CH2OH��CH3COOH
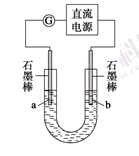
ʵ�����У���һ��Ũ�ȵ���ȩ��Na2SO4��ҺΪ�������Һ��ģ����ȩ��ˮ�Ĵ������̣���װ����ͼ��ʾ��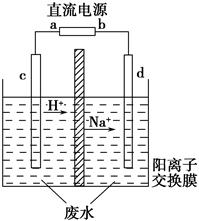
�����Լ������ȼ�ϵ��Ϊֱ����Դ����ȼ�ϵ����b��Ӧͨ��__________���ѧʽ�����缫��ӦʽΪ____________________���������У�������Na2SO4�����ʵ���________�����������С�����䡱����
����ʵ�ʹ��մ����У���������ȩ��ȥ���ʿɴ�60%�������������ֱ�ע��1 m3��ȩ����Ϊ3 000 mg��L��1�ķ�ˮ���ɵõ��Ҵ�________kg������������С�����һλ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͬѧ�����Ƶõ�CuSO4��Һ����ͼ��ʾװ�ã���������ʵ��̽����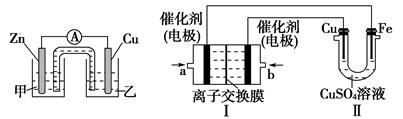
ͼһ���� ��������������������ͼ��
(1)ͼһ�Ǹ��ݷ�ӦZn��CuSO4=Cu��ZnSO4��Ƴɵ�пͭԭ��ء�Cu���ĵ缫��Ӧʽ��_____________________________________________________��
�������Ǻ���������KCl������Һ����ع���ʱK����________(��ס����ҡ�)���ƶ���
(2)ͼ���У����Ǽ���ȼ�ϵ��(�������ҺΪKOH��Һ)�Ľṹʾ��ͼ����ͬѧ���ڢ���ʵ�����϶�ͭ����b��ͨ�����________(�CH4����O2��)��a���缫�Ϸ����ĵ缫��Ӧ��_____________________________________________��
��ͭ�缫����������3.2 gʱ�����ĵ�CH4�ڱ�״���µ����Ϊ________L��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��14�֣���ҵ����ij����������Cu2O��Al2O3��Fe2O3��SiO2����ȡͭ�IJ����������£�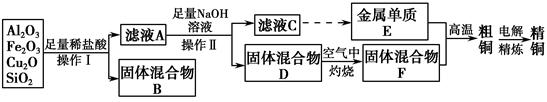
��֪��Cu2O��2H��=Cu��Cu2����H2O
��1��ʵ������������Ϊ________���ڿ��������չ�������Dʱ���õ����ֹ������ʵ������������������ƾ��ơ��������⣬����________�����������ƣ���
��2����ҺA����Ԫ�صĴ�����ʽΪ________�������ӷ��ţ������ɸ����ӵ����ӷ���ʽΪ_______________________________________________
________��������ҺA�д��ڸ����ӵ��Լ�Ϊ________�����Լ����ƣ���
��3����������E���������F������ijһ��Ӧ�����ں��Ӹֹ죬�÷�Ӧ�Ļ�ѧ����ʽΪ_____________________________________________________��
��4�������£���pH��NaAlO2��NaOH������Һ�У���ˮ�������c��OH����ǰ��Ϊ���ߵ�108������������Һ��pH��________��
��5�������õ�ⷨ���д�ͭ����ʱ������������ȷ����________������ţ���
a������ȫ��ת��Ϊ��ѧ��
b����ͭ�ӵ�Դ����������������Ӧ
c����ͭ��������������Һ��Cu2��Ũ�ȼ�С
d����ͭ����ʱͨ���ĵ�������������ͭ������ȷ����ϵ
�ڴ�Ũ���ᡢŨ���ᡢ����ˮ��ѡ�ú��ʵ��Լ����ⶨ��ͭ��Ʒ�н���ͭ�������������漰����Ҫ���裺��ȡһ����������Ʒ��______________�����ˡ�ϴ�ӡ����������ʣ�����ͭ������������ȱ�ٵIJ������裬���������������̵�ϸ�ڣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ͼ��ʾ���ձ��ж�ʢ��ϡ���ᡣ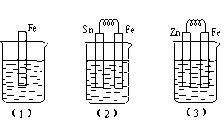
��д����3���еĵ缫��Ӧ��Fe�� �� Zn�� ��
�ڱȽϣ�1������2������3���д�������ʴ�������ɿ쵽����˳���� ������ţ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������������ɫ�ͳ����µ�Ksp���±���ʾ��
| | Cu(OH)2 | CuOH | CuCl | Cu2O |
| ��ɫ | ��ɫ | ��ɫ | ��ɫ | ש��ɫ |
| Ksp(25 ��) | 1��6��10��19 | 1��0��10��14 | 1��2��10��6 | �� |

�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com