
(1)ĻąĶ¬Ģõ¼žĻĀ£¬²āµĆ¢ŁNaClO£»¢ŚNaHCO3£»¢ŪCH3COONaČżÖÖĻ”ČÜŅŗµÄpHĻąĶ¬£®ÄĒĆ“£¬ĖüĆĒµÄĪļÖŹµÄĮæÅضČÓɓ󵽊”µÄĖ³Šņ(ŅŌĻĀ¾łĢīŠ“±ąŗÅ)________£»
(2)ÅØ¶Č¾łĪŖ0.1mol/LµÄĻĀĮŠČÜŅŗ£ŗ¢ŁHNO3£»¢ŚH2SO4£»¢ŪCH3COOH£»¢ÜBa(OH)2£»¢ŻNaOH£»¢ŽNH3”¤H2O£»¢ßNaCl£»¢ąCH3COONa£»¢įNH4Cl£¬ø÷ČÜŅŗpHÓÉŠ”µ½“óµÄĖ³ŠņŹĒ________£»
(3)ÅØ¶Č¾łĪŖ0.1mol/LµÄĻĀĮŠČÜŅŗ£ŗ¢ŁNaCl£»¢ŚNaHCO3£»¢ŪNa2CO3£»¢ÜH2SO4£»¢ŻNaOH£¬ø÷ČÜŅŗÖŠĖ®µÄµēĄė³Ģ¶ČÓÉŠ”µ½“óµÄŹĒ________£®

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
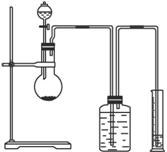
(1)Öø³öŹµŃé×°ÖƵēķĪó________________________________”£
(2)ČōŹµŃéÖŠÓĆŗ¬ÓŠÉŁĮæĖ®µÄŅŅ“¼“śĢęĻąĶ¬ÖŹĮæµÄĪŽĖ®ŅŅ“¼£¬ĻąĶ¬Ģõ¼žĻĀ£¬²āµĆĒāĘųµÄĢå»ż½«_________£ØĢī”°Ę«“ó”±”°Ę«Š””±»ņ”°²»±ä”±£©”£
(3)ĒėÖø³öÄÜŹ¹ŹµŃé°²Č«”¢Ė³Ąū½ųŠŠµÄ¹Ų¼üŹµŃé²½Öč£ØÖĮÉŁÖø³öĮ½øö¹Ų¼ü²½Öč£©
_____________________________________________________________________ӣ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
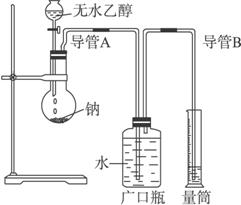
£Ø1£©Öø³öŹµŃé×°ÖĆÖŠµÄ“ķĪó____________”£
£Ø2£©ČōŹµŃéÖŠÓĆŗ¬ÓŠÉŁĮæĖ®µÄŅŅ“¼“śĢęĻąĶ¬ÖŹĮæµÄĪŽĖ®ŅŅ“¼£¬ĻąĶ¬Ģõ¼žĻĀ£¬²āµĆµÄĒāĘųĢå»ż½«____________£ØĢī”°Ę«“ó”±”°Ę«Š””±»ņ”°²»±ä”±£©”£
£Ø3£©ĒėÖø³öÄÜŹ¹øĆŹµŃé°²Č«”¢Ė³Ąū½ųŠŠµÄ¹Ų¼üŹµŃé²½Öč£ØÖĮÉŁÖø³öĮ½øö¹Ų¼ü²½Öč£©_____________________________________________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
(1)ĻņNa2CO3µÄÅØČÜŅŗÖŠÖšµĪ¼ÓČėĻ”ŃĪĖį£¬Ö±µ½²»ŌŁÉś³ÉCO2ĘųĢåĪŖÖ¹£¬ŌņŌŚ“Ė¹ż³ĢÖŠ£¬ČÜŅŗµÄc(HCO)±ä»ÆĒ÷ŹĘæÉÄÜŹĒ£ŗ¢ŁÖš½„¼õŠ”£»¢ŚÖš½„Ōö“󣻢ŪĻČÖš½„Ōö“󣬶ųŗó¼õŠ”£»¢ÜĻČÖš½„¼õŠ”£¬¶ųŗóŌö“ó”£4ÖÖĒ÷ŹĘÖŠæÉÄܵďĒ________”£
(2) pHĻąĶ¬µÄHClČÜŅŗ”¢H2SO4ČÜŅŗ”¢CH3COOHČÜŅŗø÷100 mL”£
¢Ł3ÖÖČÜŅŗÖŠ£¬ĪļÖŹµÄĮæÅضČ×ī“óµÄŹĒ________”£
¢Ś·Ö±šÓĆ0.1 mol/LµÄNaOHČÜŅŗÖŠŗĶ£¬ĘäÖŠĻūŗÄNaOHČÜŅŗ×ī¶ąµÄŹĒ________”£
¢ŪÓėµČĮæµÄŠæ·Ū·“Ó¦£¬·“Ó¦æŖŹ¼Ź±£¬·“Ó¦ĖŁĀŹ________”£
A£®HCl×īæģ B£®H2SO4×īæģ C£®CH3COOH×īæģ D£®Ņ»Ńłæģ
(3)ijĪĀ¶ČĻĀ£¬“æĖ®ÖŠc(H£«)£½2”Į10£7mol/L£¬Ōņ“ĖŹ±c(OH£)£½________mol/L”£ĻąĶ¬Ģõ¼žĻĀ£¬²āµĆ£ŗ¢ŁNaHCO3ČÜŅŗ£¬¢ŚCH3COONaČÜŅŗ£¬¢ŪNaClOČÜŅŗ£¬3ÖÖČÜŅŗµÄpHĻąĶ¬£¬ÄĒĆ“£¬ĖüĆĒµÄĪļÖŹµÄĮæÅضČÓÉŠ”µ½“óµÄĖ³ŠņŹĒ ”£
(4)ŅŃÖŖ£ŗĖįŠŌH2CO3>HClO>HCO£¬ÅŠ¶ĻŌŚµČÅØ¶ČµÄNaClO”¢NaHCO3»ģŗĻŅŗÖŠ£¬ø÷ÖÖĄė×ÓÅØ¶Č¹ŲĻµÕżČ·µÄŹĒ________
A£®c(Na£«)>c(HCO)>c(H£«)>c(OH£)
B£®c(HCO)>c(ClO£)>c(OH£)
C£®c(HClO)£«c(ClO£)£½c(HCO)£«c(CO)£«c(H2CO3)
D£®c(Na£«)£«c(H£«)£½c(HCO)£«c(ClO£)£«c(OH£)£«c(CO)
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010Äźŗ£ÄĻŹ”¼Ī»żÖŠŃ§ø߶žÉĻѧʌ½Ģѧ֏Įæ¼ģ²ā£ØČż£©»ÆѧŹŌ¾ķ ĢāŠĶ£ŗĢīæÕĢā
(1)ĻņNa2CO3µÄÅØČÜŅŗÖŠÖšµĪ¼ÓČėĻ”ŃĪĖį£¬Ö±µ½²»ŌŁÉś³ÉCO2ĘųĢåĪŖÖ¹£¬ŌņŌŚ“Ė¹ż³ĢÖŠ£¬ČÜŅŗµÄc(HCO)±ä»ÆĒ÷ŹĘæÉÄÜŹĒ£ŗ¢ŁÖš½„¼õŠ”£»¢ŚÖš½„Ōö“󣻢ŪĻČÖš½„Ōö“󣬶ųŗó¼õŠ”£»¢ÜĻČÖš½„¼õŠ”£¬¶ųŗóŌö“ó”£4ÖÖĒ÷ŹĘÖŠæÉÄܵďĒ________”£
(2) pHĻąĶ¬µÄHClČÜŅŗ”¢H2SO4ČÜŅŗ”¢CH3COOHČÜŅŗø÷100 mL”£
¢Ł3ÖÖČÜŅŗÖŠ£¬ĪļÖŹµÄĮæÅضČ×ī“óµÄŹĒ________”£
¢Ś·Ö±šÓĆ0.1 mol/LµÄNaOHČÜŅŗÖŠŗĶ£¬ĘäÖŠĻūŗÄNaOHČÜŅŗ×ī¶ąµÄŹĒ________”£
¢ŪÓėµČĮæµÄŠæ·Ū·“Ó¦£¬·“Ó¦æŖŹ¼Ź±£¬·“Ó¦ĖŁĀŹ________”£
A£®HCl×īæģ B£®H2SO4×īæģ C£®CH3COOH×īæģ D£®Ņ»Ńłæģ
(3)ijĪĀ¶ČĻĀ£¬“æĖ®ÖŠc(H£«)£½2”Į10£7mol/L£¬Ōņ“ĖŹ±c(OH£)£½________mol/L”£ĻąĶ¬Ģõ¼žĻĀ£¬²āµĆ£ŗ¢ŁNaHCO3ČÜŅŗ£¬¢ŚCH3COONaČÜŅŗ£¬¢ŪNaClOČÜŅŗ£¬3ÖÖČÜŅŗµÄpHĻąĶ¬£¬ÄĒĆ“£¬ĖüĆĒµÄĪļÖŹµÄĮæÅضČÓÉŠ”µ½“óµÄĖ³ŠņŹĒ ”£
(4)ŅŃÖŖ£ŗĖįŠŌH2CO3>HClO>HCO£¬ÅŠ¶ĻŌŚµČÅØ¶ČµÄNaClO”¢NaHCO3»ģŗĻŅŗÖŠ£¬ø÷ÖÖĄė×ÓÅØ¶Č¹ŲĻµÕżČ·µÄŹĒ________
A£®c(Na£«)>c(HCO)>c(H£«)>c(OH£)
B£®c(HCO)>c(ClO£)>c(OH£)
C£®c(HClO)£«c(ClO£)£½c(HCO)£«c(CO)£«c(H2CO3)
D£®c(Na£«)£«c(H£«)£½c(HCO)£«c(ClO£)£«c(OH£)£«c(CO)
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com