
��ͼ��ij��ҵ��Ƶ����ᡪ�ʡ�ˮ����������ˮ����ˮ���á��Ρ��ȡ���������������̬��ҵ������ͼ��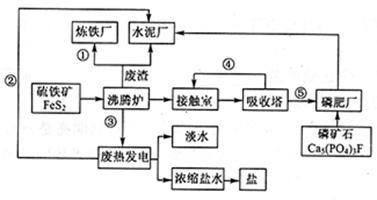
����������ҵ���̻ش��������⣺
��1�������̢١��ڡ��ۡ��ܡ���Ϊ���������ʵ����ͣ���ֱ�д�����͵���Ҫ���ʵĻ�ѧʽ��������ʽ���� ���� ���� ���� ���� ��
��2������¯������Ӧ�Ļ�ѧ����ʽ�� ���ʳ�����Ҫ��Ʒ���ո�(�������ƺ������)��д������ʯ�����ᷴӦ���ոƵû�ѧ����ʽ ��
��3����1��������(FeS2����������Ϊ36��)�Ӵ��������ᣬ������IJ���Ϊ65�������������������������Ϊ98�������� �֡�
��4���ȵ糧����ȴˮ�� ����������Ũ����ˮ����ȡ���������ȡ�������� ��д��һ�ּ��ɣ���
��5�������ִ���������������������¯�������������������������ܵ��������롣
�� ��д�����㼴�ɣ���
��1����Fe2O3 �ڵ��� ������ ��SO2 ��ŨH2SO4 ����1�֣�
��2��4FeS2+11O2 2Fe2O3+8SO2��2�֣�2Ca5(PO4)3F+7H2SO4��3Ca(H2PO4)2+7CaSO4+2HF��2�֣�
2Fe2O3+8SO2��2�֣�2Ca5(PO4)3F+7H2SO4��3Ca(H2PO4)2+7CaSO4+2HF��2�֣�
��3��0��39��2�֣� ��4����ˮ þ���� ��2�֣�
��5����������Ҫ�Ǹ�¯ú���������������Ϊȼ�ϡ� ��2�֣�
��������Ҫ�ɷ��ǹ���Ƶȣ���������ˮ������ԭ�ϡ�
��������Ҫ�ɷ��ǹ���Ƶȣ��������������ʵ�����ԭ�ϡ�
���������������1������ұ��������ԭ����Fe2O3�������ȵ糧�������ṩ������Ϊ���ܣ��۷���¯��FeS2��������Ӧ�ų��������ȣ���������ʱ�Ӵ����ж������������������Ṥҵ�����ɵ����ᣬ���������ʣ�
��2������¯��FeS2��������Ӧ����Fe2O3��SO2���䷴Ӧ����ʽΪ��4FeS2+11O2 2Fe2O3+8SO2������������ʯ��Ӧ�����������ơ�����ƣ�����ԭ���غ��֪�����з��������ɣ���˷�Ӧ�Ļ�ѧ����ʽΪ2Ca5(PO4)3F+7H2SO4��3Ca(H2PO4)2+7CaSO4+2HF��
2Fe2O3+8SO2������������ʯ��Ӧ�����������ơ�����ƣ�����ԭ���غ��֪�����з��������ɣ���˷�Ӧ�Ļ�ѧ����ʽΪ2Ca5(PO4)3F+7H2SO4��3Ca(H2PO4)2+7CaSO4+2HF��
��3������ԭ���غ��֪
FeS2��������������������2H2SO4
120t 2��98t
1t��36%��65% m��98%
��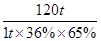 ��
��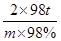
���m��0��39t
��4���غ������зḻ�ĺ�ˮ��Դ����ˮ�к��зḻ��MgԪ�ء���Ԫ�صȿ�����ȡMg���壻
��5�����ݹ����ķ����������ijɷֿ�֪����������Ҫ�Ǹ�¯ú���������������Ϊ�ȷ�¯������¯��¯��ȼ�ϣ���������Ҫ�ɷ��ǹ���Ƶȣ���������ˮ������ԭ�ϣ��ʴ�Ϊ����������Ҫ�Ǹ�¯ú���������������Ϊ�ȷ�¯������¯��¯��ȼ�ϣ���������Ҫ�ɷ��ǹ���Ƶȣ���������ˮ������ԭ�ϣ�
���㣺���鳣��������ת����ʽ�����������Ⱦ�����������ʵķ��롢�ᴿ�Ļ�������ѡ����Ӧ��


 �����ҵ���������ϵ�д�
�����ҵ���������ϵ�д� �����̸�Ӯ����ٸ�Ч�����ܸ�ϰ���ϿƼ�������ϵ�д�
�����̸�Ӯ����ٸ�Ч�����ܸ�ϰ���ϿƼ�������ϵ�д� �����ҵ�����������ѧ���ӳ�����ϵ�д�
�����ҵ�����������ѧ���ӳ�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��ˮ��Դ�ḻ����ˮ���������ѳ�Ϊ��ѧ�ҵ���Ҫ�о�������ʵ�к�ˮ��������Ӧ����ˮ�����з�������ȷ���� ( )
| A������̫���ܣ�����ˮ���� |
| B������������ʹ��ˮ���ηݳ��������� |
| C��ֱͨ���磬���õ�����ʹ��ˮ���� |
| D��ͨ�����ӽ���Ĥ����ȥ�������η�ʹ��ˮ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ú����Ҫ����Դ��Ҳ������������Ʒ����Ҫԭ�ϡ�������ѧ֪ʶ������������⣺
��1��ú��ת����������ú������������Һ��������ú��Һ�������ַ�Ϊ �� ��
��2����úȼ��ǰ���ú������������ú��ij����������ԭ������ͼ��ʾ��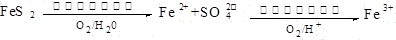
������������Ϊ�������������ü����ĵ�һ����Ӧ�����ӷ���ʽΪ ���ڶ�����Ӧ�����ӷ���ʽΪ ��
��3����ҵú����õ��IJ�Ʒ�н�̿�� ��
��4����ҵ����Ҫ���ð��������������ᣬ��ͼ�ǰ��������백������������������ȵĹ�ϵ������ֱ�߱�ʾ��Ӧ������ֵ�����߱�ʾ����ʵ����������������ʴﵽ100���������Ϧã�n(O2)��n(NH3)���� ��ʵ������Ҫ����ֵά����1.7��2.2֮�䣬 ԭ���� ��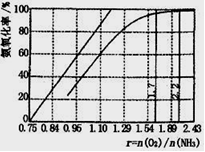
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�����������ڸ��ϲ��ϵ���
| A����ͨ���� | B����ͨ�� | C�������� | D��þ���Ͻ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ѧѡ��-��ѧ�뼼����(15��)
��ʯ�����ȼҵ�е�һ�ַ���������������±���ʾ��
�õ�ʯ����������ˮCaCl2��ij��������������¹������̣�
��֪�Ȼ��ƾ���Ļ�ѧʽ�ǣ�CaCl2��6H2O��H2S��һ���������壬�Ҿ��л�ԭ�ԡ�
�ŷ�Ӧ���м������Ӧѡ��___________________��
����ɫ����Ӧ���������X��_______________���豸A��������______________���豸B������Ϊ________________���豸C��������____________________��
��Ϊ�����㻷��Ҫ���轫����H2Sͨ�����ճأ��������������ʺ���Ϊ���ռ�����____________ _����Ӧ�Ļ�ѧ����ʽΪ_________________��
| A��ˮ | B��Ũ���� | C��ʯ���� | D������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���Ȼ���(TiCl4)����ȡ���캽�չ�ҵ���ϡ����ѺϽ����Ҫԭ�ϡ���������(��Ҫ�ɷ���FeTiO3)�Ʊ�TiCl4�Ȳ�Ʒ��һ�ֹ�������ʾ��ͼ���£�
(1)�����м�����м������Һ����ɫ����ʱ��Һ�Գ�ǿ���ԡ��ù����������·�Ӧ������
Fe+2Fe3+=3Fe2+
2TiO2+(��ɫ)+Fe+4H+=2Ti3+(��ɫ)+Fe2++2H2O
Ti3+(��ɫ)+Fe3++H2O=TiO2+(��ɫ)+Fe2++2H+
������������� ��
(2)�ڢڡ� �۹��չ�������Ҫ�����������γ�TiO2��nH2O�ܽ������ܽ��ķ�ɢ�ʿ���ֱ����С�� ��Χ��
(3)���Ѣ����ƵõĹ���TiO2��nH2O������ϴ��ȥ���е����ʣ������Ƶ��Ѱۡ���֪25��ʱ��Ksp��Fe(OH)3��=2.79��10-39�����¶��·�ӦFe(OH)3+3H+  Fe3++3H2O��ƽ�ⳣ��K= ��
Fe3++3H2O��ƽ�ⳣ��K= ��
(4)��֪��TiO2(s)+2Cl2(g)=TiCl4(l)+O2(g)
��H="+140" kJ��mol��1
2C(s)+O2(g)=2CO(g) ��H����221 kJ��mol��1
д������TiO2�ͽ�̿��������Ӧ����Һ̬TiCl4��CO������Ȼ�ѧ����ʽ�� ��
(5)�������վ��гɱ��͡����õ�Ʒλ����Ϊԭ�ϵ��ŵ㡣������ɫ��ѧ����ù��������д��ڵIJ���֮���� (ֻҪ��д��һ�� )��
(6)�����±���Ϣ��Ҫ���ƺ�����SiCl4���ʵ�TiCl4���ɲ���_______������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
̼��﮹㷺Ӧ�����մɺ�ҽҩ��������﮻�ʯ����Ҫ�ɷ�ΪLiAlSi2O6��Ϊԭ�����Ʊ�Li2CO3�Ĺ����������£�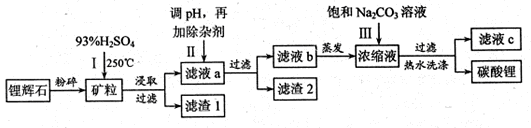
��֪��2LiAlSi2O6+H2SO4 Li2SO4+Al2O3·4SiO2?H2O
Li2SO4+Al2O3·4SiO2?H2O
��Fe3+��Al3+��Fe2+��Mg2+������������ʽ��ȫ����ʱ����Һ��PH�ֱ�Ϊ3.2��4.7��9.0��11.1
��ijЩ���ʵ��ܽ�ȣ�S�����±�
��ش��������⣺
��1��﮻�ʯ��Ũ�����ȡ֮ǰҪ�����ϸ������Ŀ���� ��
��2����Һa�к���Li+��SO42-,������Fe3+��Al3+��Fe2+�� Mg2+ ��Ca2+ ��Na+�����ʣ���������ڽ����¼���ʯ��ʯ�Ե�����Һ��PH��6.0��6.5����ʱ���������������� ��ʯ��ʯ������ҺPH��ԭ�������ӷ���ʽΪ ��
��3�����������Һa�м���ij��Ӽ�����Ϊ������H2O2��Һ��ʯ�����Na2CO3��Һ������������ԭ��Ӧ�����ӷ���ʽΪ ��
��4��������м��뱥��Na2CO3��Һ���˺���Ҫ����ˮϴ�ӵ�ԭ���� ��
��5������Һc�пɻ��յ���Ҫ������ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������(K2Fe04)��һ�ּ�������������������һ������Ͷ��ˮ���������������������£�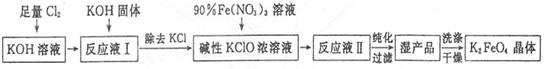
��֪��2Fe(NO3)3+3KClO+10KOH=2K2FeO4+6KNO3+3KCl+5H2O
�ش�����������
��1����Cl2ͨ��KOH��Һ�з�����Ӧ�����ӷ���ʽ��____________��
��2��д����ҵ����ȡCl2�Ļ�ѧ����ʽ____________��
��3���ڡ���ӦҺI���м���KOH�����Ŀ����____________��
��4��K2FeO4����Ϊ���Ͷ��ˮ��������ԭ����____________��
��5������KOH��Һʱ����61.6g KOH�����ܽ���100 mLˮ�У�������Һ���ܶ�Ϊ1.47 g ? mL-1�������Һ�����ʵ���Ũ��Ϊ____________��
��6���ӡ���ӦҺII���з����K2Fe04����Ʒ��___________ (д�� ѧ ʽ����
��7���ù���ÿ�õ�1.98kgK2FeO4������������Cl2�����ʵ���Ϊ______mol��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
ij������CaSO4��NH3��H2O��CO2�Ʊ�(NH4)2SO4���乤���������£�
�����ƶϲ��������� (����)��
| A������1 mol(NH4)2SO4��������2 mol NH3 |
| B��CO2�ɱ�ѭ��ʹ�� |
| C��������ͨCO2������(NH4)2SO4������ |
| D��ֱ��������Һ���ɵõ�������(NH4)2SO4 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com