
(1)�û�ѧ����ʽ�и����ʵĻ�ѧ������Ϊ��
a=_____________��b=_____________��c=_____________��d=____________��
(2)��ֻ�ı�ѹǿ����Ӧ���ʷ����仯����ƽ�ⲻ�����ƶ����÷�Ӧ�и����ʵľۼ�״̬��
A_____________��B_____________��C_____________��D_____________��
(3)��ֻ�����¶ȣ���Ӧһ��ʱ�����������ʵ����ʵ����ִﵽ��ȣ���÷�ӦΪ_____________��Ӧ(����ȡ������ȡ�)��
������(1)��仯�����ʵ���֮�ȵ��ڻ�ѧ�������ȣ�����a��b��c��d��n��![]() ��
��![]() n��n��2��1��3��2����a=2��b=1��c=3��d=2��
n��n��2��1��3��2����a=2��b=1��c=3��d=2��
(2)��ı�ѹǿ�����ʷ����仯˵������̬���ʲμӷ�Ӧ��ʹƽ�ⲻ�ƶ���˵���Ⱥ�������̬���ʵĻ�ѧ��������ȣ��ݴ˿���֪��AΪ��̬��BΪ��̬��Һ̬��CΪ��̬��DΪ��̬��
(3)����ʱ����Ӧһ��ʱ����������ʵ����ִﵽ��ȣ�˵��ƽ�����淴Ӧ�����ƶ��������淴ӦΪ���ȷ�Ӧ��
�𰸣�(1)2 1 3 2
(2)��̬ ��̬��Һ̬ ��̬ ��̬
(3)����


 ��У���˳�̾�ϵ�д�
��У���˳�̾�ϵ�д� ��У���һ��ͨϵ�д�
��У���һ��ͨϵ�д� �γ̴����Ծ�����100��ϵ�д�
�γ̴����Ծ�����100��ϵ�д� �¾�����ĩ���100��ϵ�д�
�¾�����ĩ���100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ������ʡ����һ�и�һ��ѧ����ĩ���Ի�ѧ�Ծ����������� ���ͣ�������
��8�֣��������ʵ�����A��B�����1 L���ܱ������У��������·�Ӧ
3A(g)��B(g)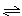 xC(g)��2D(g)����5 min���D��Ũ��Ϊ0.5 mol/L��c(A)��c(B)��3��5��C��ƽ����Ӧ����Ϊ0.1 mol/(L��min)����
xC(g)��2D(g)����5 min���D��Ũ��Ϊ0.5 mol/L��c(A)��c(B)��3��5��C��ƽ����Ӧ����Ϊ0.1 mol/(L��min)����
(1)��ʱA��Ũ��c(A)��________mol/L����Ӧ��ʼǰ�����е�A��B�����ʵ�����n(A)��n(B)��________mol��
(2)B��ƽ����Ӧ����v(B)��________mol/(L��min)��
(3)x��ֵΪ________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014������ʡ��һ��ѧ����ĩ���Ի�ѧ�Ծ��������棩 ���ͣ�������
��8�֣��������ʵ�����A��B�����1 L���ܱ������У��������·�Ӧ
3A(g)��B(g) 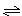 xC(g)��2D(g)����5 min���D��Ũ��Ϊ0.5 mol/L��c(A)��c(B)��3��5��C��ƽ����Ӧ����Ϊ0.1 mol/(L��min)����
xC(g)��2D(g)����5 min���D��Ũ��Ϊ0.5 mol/L��c(A)��c(B)��3��5��C��ƽ����Ӧ����Ϊ0.1 mol/(L��min)����
(1)��ʱA��Ũ��c(A)��________mol/L����Ӧ��ʼǰ�����е�A��B�����ʵ�����n(A)��n(B)��________mol��
(2)B��ƽ����Ӧ����v(B)��________mol/(L��min)��
(3)x��ֵΪ________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�콭��ʡ��һ��ѧ�ڵ����ζο���ѧ�Ծ� ���ͣ������
(4��)�������ʵ�����A��B�����2 L���ܱ������У��������·�Ӧ3A(g)��B(g)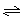 xC(g)��2D(g)����5 min���D��Ũ��Ϊ0.5 mol/L��c(A)��c(B)��3��5��C��ƽ����Ӧ����Ϊ0.1 mol/(L��min)����
xC(g)��2D(g)����5 min���D��Ũ��Ϊ0.5 mol/L��c(A)��c(B)��3��5��C��ƽ����Ӧ����Ϊ0.1 mol/(L��min)����
(1)��ʱA��Ũ��c(A)�� mol/L����Ӧ��ʼǰ�����е�A��B�����ʵ�����n(A)��n(B)�� mol��
(2)B��ƽ����Ӧ����v(B)�� mol/(L��min)�� (3)x��ֵΪ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com