

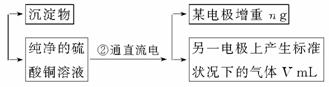
»Ų“šĻĀĮŠĪŹĢā”£
(1)¼ÓČėCuOµÄ×÷ÓĆŹĒ _______________________________ ”£
(2)²½Öč¢ŚÖŠĖłÓĆµÄ²æ·ÖŅĒĘ÷ČēĻĀĶ¼ĖłŹ¾£¬ŌņA”¢B·Ö±šĮ¬Ö±Į÷µēŌ“µÄ¼«_______ŗĶ_______¼«(Ģī”°Õż”±»ņ”°øŗ”±)
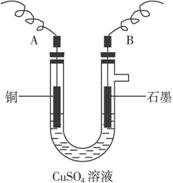
(3)µē½āæŖŹ¼ŗó£¬ŌŚUŠĪ¹ÜÖŠæÉŅŌ¹Ū²ģµ½µÄĻÖĻóÓŠ£ŗ________________”£µē½āµÄĄė×Ó·½³ĢŹ½ĪŖ________________________________________________”£
(4)ĻĀĮŠŹµŃé²Ł×÷ÖŠ±ŲŅŖµÄŹĒ_______ (Ģī×ÖÄø)”£
A.³ĘĮæµē½āĒ°µē¼«µÄÖŹĮæ
B.µē½āŗ󣬵ē¼«ŌŚŗęøɳĘĮæĒ°£¬±ŲŠėÓĆÕōĮóĖ®³åĻ“
C.¹ĪĻĀµē½āŗóµē¼«ÉĻĪö³öµÄĶ£¬²¢ĒåĻ“”¢³ĘĮæ
D.µē½āŗóŗęøɳĘÖŲµÄ²Ł×÷ÖŠ±ŲŠė°“”°ŗęøɳĘĮæŌŁŗęøÉŌŁ³ĘĮæ”±½ųŠŠ
E.ŌŚÓŠæÕĘų“ęŌŚµÄĒéæöĻĀ£¬ŗęøɵē¼«±ŲŠėÓƵĶĪĀŗęøɵķ½·Ø
(5)ĶµÄĻą¶ŌŌ×ÓÖŹĮæĪŖ__________________ (ÓĆ“ųÓŠm”¢VµÄ¼ĘĖćŹ½±ķŹ¾)”£
½āĪö£ŗ±¾ĢāŹĒŅ»µĄ¶ØĮæŹµŃé×ŪŗĻĢā£¬ÖŠŠÄŹĒ×¼Č·²āĮæ³öCuµÄÖŹĮæ¼°µē¼«ÉĻ²śÉśµÄĘųĢåĢå»ż£¬“ÓĢāøÉÖŖ£¬pHĪŖ4”Ŗ5µÄĢõ¼žĻĀ£¬Cu2+¼øŗõ²»Ė®½ā£¬¶ųFe3+¼øŗõĶźČ«Ė®½ā£¬ĖłŅŌ³żCuSO4ČÜŅŗÖŠµÄFe(SO4)3ČÜŅŗ£¬æÉĄūÓĆÉĻŹöŠÅĻ¢£¬¼ÓČėCuO¾ĶŹĒĻūŗÄH+,¶ųŹ¹Fe3+µÄĖ®½āĘ½ŗāĻņÓŅŅĘ¶Æ£¬Ź¹Fe3+ĶźČ«Ė®½āŠĪ³ÉFe(OH)3³Įµķ¶ų³żČ„£»ÓÉÓŚ±¾ĢāŹĒµē½āCuSO4ČÜŅŗ£¬Ņņ“Ėµē¼«µÄĮ¬½Ó¼°ĻÖĻóµÄĆčŹö¾łæÉøł¾Żµē½āµÄ×Ü»Æѧ·½³ĢŹ½£¬2CuSO4+2H2O![]() 2Cu+O2”ü+2H2SO4£¬¶ųŅŖ×¼Č·²ā¶ØĪö³öCuµÄÖŹĮ棬¾ĶŠčŃĻøń°“ÕÕŹµŃé²Ł×÷£¬A”¢B”¢D”¢EĻī²Ł×÷ŹĒ²»æÉȱɣµÄ£»ÓŠĮĖCuµÄÖŹĮæ¼°O2ŌŚ±ź×¼×“æöĻĀµÄĢå»ż£¬øł¾Ż·½³ĢŹ½¼“æɼĘĖć³öCuµÄĻą¶ŌŌ×ÓÖŹĮæĪŖ
2Cu+O2”ü+2H2SO4£¬¶ųŅŖ×¼Č·²ā¶ØĪö³öCuµÄÖŹĮ棬¾ĶŠčŃĻøń°“ÕÕŹµŃé²Ł×÷£¬A”¢B”¢D”¢EĻī²Ł×÷ŹĒ²»æÉȱɣµÄ£»ÓŠĮĖCuµÄÖŹĮæ¼°O2ŌŚ±ź×¼×“æöĻĀµÄĢå»ż£¬øł¾Ż·½³ĢŹ½¼“æɼĘĖć³öCuµÄĻą¶ŌŌ×ÓÖŹĮæĪŖ![]() ”£
ӣ
“š°ø£ŗ(1)ĶعżĻūŗÄH+¶ųµ÷ÕūČÜŅŗµÄpHŹ¹Ö®Éżøߣ¬ĘäÄæµÄŹĒŹ¹Fe3+ĶźČ«Ė®½āŠĪ³ÉFe(OH)3³Įµķ¶ų³żČ„
(2)øŗ Õż
(3)A¼«ÉĻÓŠŗģÉ«ĪļÖŹĪö³ö£¬B¼«ÉĻÓŠĘųÅŻŅŻ³ö£¬ČÜŅŗĄ¶É«±äĒ³
2Cu2++2H2O![]() 2Cu+4H++O2ӟ
2Cu+4H++O2ӟ
(4)AӢBӢDӢE (5) ![]()


 ½šÅĘ½ĢøØÅąÓÅÓÅŃ”¾ķĘŚÄ©³å“Ģ100·ÖĻµĮŠ“š°ø
½šÅĘ½ĢøØÅąÓÅÓÅŃ”¾ķĘŚÄ©³å“Ģ100·ÖĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā

| ³ĮµķĪļ | Fe£ØOH£©3 | Al£ØOH£©3 | Mg£ØOH£©2 |
| pH | 3.4 | 5.2 | 12.4 |

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
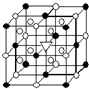 A£®£Ø1£©ÓŅĶ¼ĖłŹ¾ĪŖ±ł¾§ŹÆ£Ø»ÆѧŹ½ĪŖNa3AlF6£©µÄ¾§°ū£®Ķ¼ÖŠ”ńĪ»ÓŚ“óĮ¢·½Ģ嶄µćŗĶĆęŠÄ£¬”šĪ»ÓŚ“óĮ¢·½ĢåµÄ12ĢõĄāµÄÖŠµćŗĶ8øöŠ”Į¢·½ĢåµÄĢåŠÄ£¬ØĶ¼ÖŠ”ń”¢”šÖŠµÄŅ»ÖÖ£®Ķ¼ÖŠ”ń”¢”š·Ö±šÖø“śÄÄÖÖĮ£×Ó
A£®£Ø1£©ÓŅĶ¼ĖłŹ¾ĪŖ±ł¾§ŹÆ£Ø»ÆѧŹ½ĪŖNa3AlF6£©µÄ¾§°ū£®Ķ¼ÖŠ”ńĪ»ÓŚ“óĮ¢·½Ģ嶄µćŗĶĆęŠÄ£¬”šĪ»ÓŚ“óĮ¢·½ĢåµÄ12ĢõĄāµÄÖŠµćŗĶ8øöŠ”Į¢·½ĢåµÄĢåŠÄ£¬ØĶ¼ÖŠ”ń”¢”šÖŠµÄŅ»ÖÖ£®Ķ¼ÖŠ”ń”¢”š·Ö±šÖø“śÄÄÖÖĮ£×Ó| ČŪµć/K | ·Šµć/K | ±ź×¼×“æöŹ±ŌŚĖ®ÖŠµÄČܽā¶Č | |
| H2S | 187 | 202 | 2.6 |
| H2O2 | 272 | 423 | ŅŌČĪŅā±Č»„ČÜ |
| 80m-135n |
| 18n |
| 80m-135n |
| 18n |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

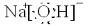
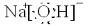
| ||
| ||
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
£Ø20·Ö£©¾ŪŗĻĀČ»ÆĀĮŹĒŅ»ÖÖŠĀŠĶ”¢øߊ§ŠõÄż¼ĮŗĶ¾»Ė®¼Į£¬Ę䵄ĢåŹĒŅŗĢ¬µÄ¼īŹ½ĀČ»ÆĀĮ[Al2£ØOH£©nCl6-n]”£±¾ŹµŃé²ÉÓĆĀĮŃĪČÜŅŗĖ®½āŠõÄż·ØÖʱø¼īŹ½ĀČ»ÆĀĮ”£ĘäÖʱøŌĮĻĪŖ·Ö²¼¹ć”¢¼ŪøńĮ®µÄøßĮė ĶĮ£¬»Æѧ×é³ÉĪŖ£ŗAl2O3£Ø25%”«34%£©”¢SiO2£Ø40%”«50%£©”¢Fe2O3£Ø0£®5%”«3£®0%£©ŅŌ¼°ÉŁĮæŌÓÖŹŗĶĖ®·Ö”£ŅŃÖŖŃõ»ÆĀĮÓŠ¶ąÖÖ²»Ķ¬µÄ½į¹¹£¬»ÆѧŠŌÖŹŅ²ÓŠ²īŅģ£¬ĒŅŅ»¶ØĢõ¼žĻĀæÉĻą»„×Ŗ»Æ£»øßĮėĶĮÖŠµÄŃõ»ÆĀĮÄŃČÜÓŚĖį”£Öʱø¼īŹ½ĀČ»ÆĀĮµÄŹµŃéĮ÷³ĢČēĻĀ£ŗ
ŅŃÖŖ£ŗFe3£«”¢Al3£«ŅŌĒāŃõ»ÆĪļŠĪŹ½ĶźČ«³ĮµķŹ±£¬ČÜŅŗµÄpH·Ö±šĪŖ3.2”¢5.2”£
øł¾ŻĮ÷³ĢĶ¼»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©”°ģŃÉÕ”±µÄÄæµÄŹĒ_______________________________________________”£
£Ø2£©”°½ž³ö”±¹ż³ĢÖŠ·¢Éś·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ_______________________________”£
£Ø3£©”°½ž³ö”±Ń”ÓƵÄĖįĪŖ_______”£ÅäÖĘÖŹĮæ·ÖŹż15£„µÄAĖįŠčŅŖ200mL30£„µÄAĖį£ØĆܶČŌ¼ĪŖ1.15g/cm3£©ŗĶ_______gÕōĮóĖ®£¬ÅäÖĘÓƵ½µÄŅĒĘ÷ÓŠÉÕ±”¢²£Į§°ō”¢______________”£
£Ø4£©ĪŖĢįøßĀĮµÄ½ž³öĀŹ£¬æɲÉČ”µÄ“ėŹ©ÓŠ _______________£ØŅŖĒ󊓳öČżĢõ£©”£
£Ø5£©”°µ÷½ŚČÜŅŗpHŌŚ4£®2”«4£®5”±µÄ¹ż³ĢÖŠ£¬³żĢķ¼Ó±ŲŅŖµÄŹŌ¼Į£¬»¹Šč½čÖśµÄŹµŃéÓĆĘ·ŹĒ_________________£»”°Õō·¢ÅØĖõ”±Šč±£³ÖĪĀ¶ČŌŚ90”«100”ę£¬æŲÖĘĪĀ¶ČµÄŹµŃé·½·ØŹĒ___________ ______”£
£Ø6£©ŹµŃéÖŠÖʱø¼īŹ½ĀČ»ÆĀĮ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ_________________
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012½ģ½ĖÕŹ”Ńļ֯֊ѧøßČż3ŌĀĖ«ÖÜĮ·Ļ°£Ø¶ž£©»ÆѧŹŌ¾ķ£Ø“ų½āĪö£© ĢāŠĶ£ŗĢīæÕĢā
£Ø20·Ö£©¾ŪŗĻĀČ»ÆĀĮŹĒŅ»ÖÖŠĀŠĶ”¢øߊ§ŠõÄż¼ĮŗĶ¾»Ė®¼Į£¬Ę䵄ĢåŹĒŅŗĢ¬µÄ¼īŹ½ĀČ»ÆĀĮ[Al2£ØOH£©nCl6-n]”£±¾ŹµŃé²ÉÓĆĀĮŃĪČÜŅŗĖ®½āŠõÄż·ØÖʱø¼īŹ½ĀČ»ÆĀĮ”£ĘäÖʱøŌĮĻĪŖ·Ö²¼¹ć”¢¼ŪøńĮ®µÄøßĮėĶĮ£¬»Æѧ×é³ÉĪŖ£ŗAl2O3£Ø25%”«34%£©”¢SiO2£Ø40%”«50%£©”¢Fe2O3£Ø0£®5%”«3£®0%£©ŅŌ¼°ÉŁĮæŌÓÖŹŗĶĖ®·Ö”£ŅŃÖŖŃõ»ÆĀĮÓŠ¶ąÖÖ²»Ķ¬µÄ½į¹¹£¬»ÆѧŠŌÖŹŅ²ÓŠ²īŅģ£¬ĒŅŅ»¶ØĢõ¼žĻĀæÉĻą»„×Ŗ»Æ£»øßĮėĶĮÖŠµÄŃõ»ÆĀĮÄŃČÜÓŚĖį”£Öʱø¼īŹ½ĀČ»ÆĀĮµÄŹµŃéĮ÷³ĢČēĻĀ£ŗ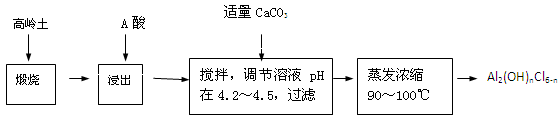
ŅŃÖŖ£ŗFe3£«”¢Al3£«ŅŌĒāŃõ»ÆĪļŠĪŹ½ĶźČ«³ĮµķŹ±£¬ČÜŅŗµÄpH·Ö±šĪŖ3.2”¢5.2”£
øł¾ŻĮ÷³ĢĶ¼»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©”°ģŃÉÕ”±µÄÄæµÄŹĒ_______________________________________________”£
£Ø2£©”°½ž³ö”±¹ż³ĢÖŠ·¢Éś·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ_______________________________”£
£Ø3£©”°½ž³ö”±Ń”ÓƵÄĖįĪŖ_______”£ÅäÖĘÖŹĮæ·ÖŹż15£„µÄAĖįŠčŅŖ200mL30£„µÄAĖį£ØĆܶČŌ¼ĪŖ1.15g/cm3£©ŗĶ_______gÕōĮóĖ®£¬ÅäÖĘÓƵ½µÄŅĒĘ÷ÓŠÉÕ±”¢²£Į§°ō”¢______________”£
£Ø4£©ĪŖĢįøßĀĮµÄ½ž³öĀŹ£¬æɲÉČ”µÄ“ėŹ©ÓŠ _______________£ØŅŖĒ󊓳öČżĢõ£©”£
£Ø5£©”°µ÷½ŚČÜŅŗpHŌŚ4£®2”«4£®5”±µÄ¹ż³ĢÖŠ£¬³żĢķ¼Ó±ŲŅŖµÄŹŌ¼Į£¬»¹Šč½čÖśµÄŹµŃéÓĆĘ·ŹĒ_________________£»”°Õō·¢ÅØĖõ”±Šč±£³ÖĪĀ¶ČŌŚ90”«100”ę£¬æŲÖĘĪĀ¶ČµÄŹµŃé·½·ØŹĒ___________ ______”£
£Ø6£©ŹµŃéÖŠÖʱø¼īŹ½ĀČ»ÆĀĮ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ_________________
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com