���ڹ�����ռ����Ҫ�ĵ�λ�����������̽����
��1��������������ˮ����ͨ�����ȵ�̿������ˮú����
C��s��+H
2O��g��?H
2��g��+CO��g����H=+131.3kJ����S=+133.7J/K
�÷�Ӧ�ڵ������ܷ��Է�
����ܻ��
��2����֪��400��ʱ��N
2 ��g��+3H
2��g��?2NH
3��g����K=0.5��
��2NH
3��g��?N
2 ��g��+3H
2��g����K=
������ֵ����
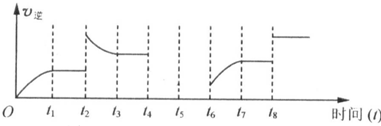
��400��ʱ����0.5L�ķ�Ӧ�����н��кϳɰ���Ӧ��һ��ʱ����N
2��H
2��NH
3�����ʵ����ֱ�Ϊ2mol��1mol��2mol�����ʱ��ӦV��N
2��
��
V��N
2��
�������������=������ȷ����
��3����������ͬ�����и�����1molN
2��3molH
2����ijһ��ͬ�����·�Ӧ���ﵽƽ�⣬�������������ʱ��仯������ͼ������˵����ȷ����
������ţ���
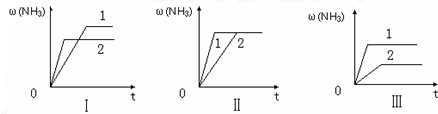
A��ͼ������Dz�ͬѹǿ�Է�Ӧ��Ӱ�죬��P
2��P
1B��ͼ������Dz�ͬѹǿ�Է�Ӧ��Ӱ�죬��P
1��P
2C��ͼ������Dz�ͬ�¶ȶԷ�Ӧ��Ӱ�죬��T
1��T
2D��ͼ�������ͬ��ͬѹ�£��������ܣ�1��2��





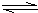 2NH3�ġ�H
2NH3�ġ�H