
��14�֣��к�����ָ�������кͷ�Ӧ����lmol H2O���ų���������ijѧ����ͨ���ⶨ��Ӧ���������ų��������������к��ȡ�����50mL0.5mol/L��������50mL0.55mol/L��NaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ����ش��������⣺
��1����ʵ��װ���Ͽ���ͼ����ȱ�ٵ�һ�ֲ�����Ʒ��______���ձ���������������ĭ��������______��
��2�����ձ����粻��Ӳֽ�壬����õ��к�����ֵ_________(�ƫ����ƫС��������Ӱ�족)
��3��ʵ���и���60 mL 0.50 mol/L�������50mL 0.55 mol/L��NaOH��Һ����Ӧ��������ʵ����ȣ����ų�������________(���ȡ�����ȡ�������________________�������к��ȵ���ֵ��________(���ȡ�����ȡ�)��������_______________________��
��14�֣���1�����β���������� ���¡�����,����ʵ������е�������ʧ
��2��ƫС
��3������ȣ� ��Ϊ�ᡢ����кͷ�Ӧ�ų����������ᡢ��������йأ� ���
�к�����ָ�������кͷ�Ӧ����lmol H2O���ų�������,�����ᡢ���������
���������������1��Ϊ��ʹ����Ͼ��ȣ��軷�β�����������裬ͬʱΪ�˼���ʵ������е�������ʧ��Ӧ���¡����ȣ����ձ���������������ĭ����2�� ���ձ����粻��Ӳֽ�壬������ʧ����õ��к�����ֵƫС����3����Ϊ�ᡢ����кͷ�Ӧ�ų����������ᡢ��������йأ�ǰ����0.5mol/L��0.050L=0.025mol���������кͷ�Ӧ��������0.55mol/L��0.050L=0.0275mol���������кͷ�Ӧ���ų�����������ȡ������к�����ָ�������кͷ�Ӧ����lmol H2O���ų�������,�����ᡢ��������أ������к�����ֵ��ȡ�
���㣺�к��ȵIJⶨ


 Сѧ���AB��ϵ�д�
Сѧ���AB��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��ѧ������������������ء�����˵���������
| A������������ʹ֯����ɫ���Խ�������Ҳ�и�ʴ���� |
| B����������������ȸ������ɼ���Ҳ������θ������һ��ҩ�� |
| C����ˮ��ʯ��������ΪCaCO3��CO2��ˮ�����˻�ѧ��Ӧ |
| D����Ϊ����Һ����������ʱ�����մ��������������Կ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���������У����������������ӵĺ�����Ӳ�����ȵ���
| A��Al3+ | B��Mg2+ | C��Na+ | D��H+ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���ڻ�ѧ���������У���ȷ����
| A�����ӻ�������ܺ����ۼ� |
| B�����ۻ�������ܺ����Ӽ� |
| C�����ӻ�������ֻ�����Ӽ� |
| D�����ӻ�������һ�����н������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijʵ��С����0��50 mol/L NaOH��Һ��0��50 mol/L H2SO4��Һ�����к��ȵIJⶨ��
������0��50 mol/L NaOH��Һ
��ʵ���д�ԼҪʹ��245 mL NaOH��Һ����������Ҫ����NaOH����________g��
�ⶨ�к��ȵ�ʵ��װ����ͼ��ʾ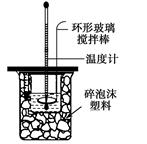
(1)д��ϡ�����ϡ����������Һ��Ӧ��ʾ�к��ȵ��Ȼ�ѧ����ʽ(�к�����ֵ57��3 kJ/mol)��________ ��
(2)ȡ50 mL NaOH��Һ��30 mL�������ʵ�飬ʵ���������±���
������д�±��еĿհף�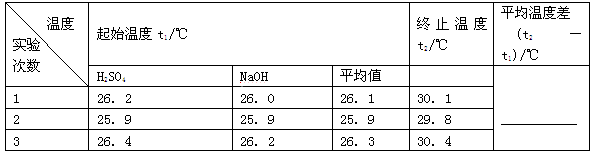
�ڽ�����Ϊ0��50 mol/L NaOH��Һ��0��50 mol/L H2SO4��Һ���ܶȶ���1 g/cm3���кͺ�������Һ�ı�����c��4��18 J/(g����)�����к��Ȧ�H��________(ȡС�����һλ)��
������ʵ��������ֵ��57��3 kJ/mol��ƫ�����ƫ���ԭ�������(����ĸ)________��
a��ʵ��װ�ñ��¡�����Ч����
b������ȡNaOH��Һ�����ʱ���Ӷ���
c���ֶ�ΰ�NaOH��Һ����ʢ��ϡ�����С�ձ���
d�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨH2SO4��Һ���¶�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��15�֣�ijʵ��С����0.50 mol/L NaOH��Һ��0.50 mol/L������Һ�����к��ȵIJⶨ��
������0.50 mol/L NaOH��Һ
��1����ʵ���д�ԼҪʹ��245 mL NaOH��Һ��������Ҫ����NaOH���� g��
��2�����ƹ�������Ҫ�õ��IJ����������ձ������������Ҫ�� �� ��
�ⶨ�к��ȵ�ʵ��װ������ͼ��ʾ��
��3����С�ձ�֮����������ĭ���ϵ������� ����ʵ��װ���Ͽ���ͼ��ȱ�ٵ�һ�ֲ������� ��
��4��ʹ�ò�ȫ�������װ�ý���ʵ�飬ȡ50mL 0.25mol��L H2SO4��Һ��50mL0.55 mol��L NaOH��Һ��С�ձ��н����кͷ�Ӧ��ʵ���������±���
������д�±��еĿհף�
| ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�¶� t2/�� | �¶Ȳ�ƽ��ֵ ��t2-t1��/�� | ||
| H2SO4 | NaOH | ƽ��ֵ | |||
| 1 | 26.2 | 26.0 | 26.1 | 29.5 | �� |
| 2 | 27.0 | 27.4 | 27.2 | 33.3 | |
| 3 | 25.9 | 25.9 | 25.9 | 29.2 | |
| 4 | 26.4 | 26.2 | 26.3 | 29.8 | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��������þ������ˮ�дﵽ�ܽ�ƽ��Mg(OH)2(s)  Mg2��(aq)��2OH��(aq)ʱ��ΪʹMg(OH)2����������٣������������
Mg2��(aq)��2OH��(aq)ʱ��ΪʹMg(OH)2����������٣������������
| A��MgCl2 | B��NaOH | C��MgSO4 | D��NaHSO4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��14�֣�I������ͼ��ʾ,50mL0��50mol��L-1������50mL0��55mol��L-1NaOH��Һ��װ���н�����
�ͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų�������,�ɼ����к���,�ش���������:
��1��ͼ��װ��ȱ�ٵ������� ��
��2������˵������ȷ���� (�����)��
A������50mL0��55mol��L-1Ba(OH)2��Һ����NaOH��Һ����ʵ��,��õ��к�����ͬ
B������50mL0��50mol��L-1������Һ�����������ʵ��,��õ��к�����ͬ
C�����ֱ��õ�Ũ�ȵ������Ba(OH)2��Һ���������NaOH��Һ����ʵ��,��õ��к�����ͬ
D��NaOH��Һ�Թ�����ԭ������������ȫ��Ӧ
II��ijѧ����0��1mol/L KOH��Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����
A����ȡ20��00mL�����������Һע��ྻ����ƿ��������2-3�η�̪
B���ñ���Һ��ϴ�ζ���2-3��
C����ʢ�б���Һ�ļ�ʽ�ζ��̶ܹ��ã�����Һ��ʹ�ζ��ܼ��������Һ
D��ȡ��KOH��Һע���ʽ�ζ�����0�̶�����2-3cm
E������Һ����0��0�̶����£����¶���
F������ƿ���ڵζ��ܵ����棬�ñ�KOH��Һ�ζ����յ㣬���µζ���Һ��Ŀ̶�
���������գ�
��1����ȷ������˳���ǣ��������ĸ��д��______________________��
��2��ʵ�����۾�ע��_______________��ֱ���ζ��յ㡣�жϵ����յ��������__________________��
��3��������ʵ������KOH��Һ����ֱ�Ϊ14��98 mL��16��80 mL��15��02 mL��c��HCl��= ��
��4�����в����У�������ɲⶨ���ƫ�ߵ��� ��
A��δ�ñ�Һ��ϴ��ʽ�ζ���
B���ζ��յ����ʱ�����ӵζ�����Һ��̶ȣ�������������ȷ
C��ʢװ����Һ����ƿ������ˮϴ����δ�ô���Һ��ϴ
D���ζ����յ����ʱ���ֵζ��ܼ��촦����һ����Һ
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com