
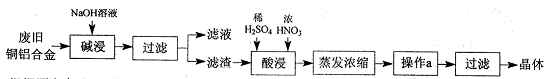


 �߲������Ӧ��һ��ͨϵ�д�
�߲������Ӧ��һ��ͨϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
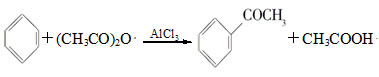
| A����Ӧ̫���� | B��Һ��̫������� | C����Ӧ�仺�� | D������������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ֽ����ʵ������ѡ��ˮ���̶��࣬�л��ܼ��������࣬����ˮ��ǿ�ijɷ����������з������һЩ����ëϸ���������£����������ƶ����ٶȿ�һЩ |
| B���ж�CuO��MnO2��H2O2�ֽ�Ĵ�Ч�ʣ���ͨ���Ƚ�H2O2��Һ�������ݵĿ����̶ȣ���ⶨ��λʱ���ڲ������������ |
| C��ʳ�����Ậ���ⶨʵ���У�ʵ�����ʱ��ʽ�ζ��ܼ��첿λ�����ݣ���ⶨ�Ĵ��Ậ��ƫ�� |
| D������������������淋Ļ����Һ����Ũ������ȴ�ᾧ�������Ħ���δֲ�Ʒ�����þƾ�ϴȥ��������ˮ�֣����Ⱥ�ɺ���أ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ɫʯ����ֽ | B����ɫʯ����ֽ | C������KI��ֽ | D��pH��ֽ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
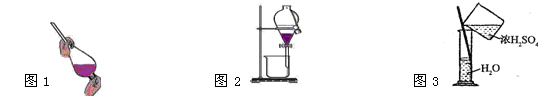
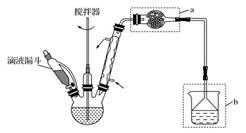
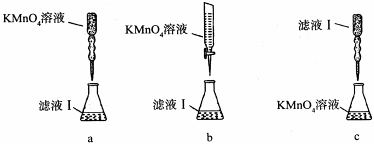
| A����ȡ����ʱ����������Ҫ������������ͨ����Һ©���ϿڵIJ��������е� |
| B�����Բ�������Ũ������ȴ�ᾧ�ķ������������Һ�л������ؾ��� |
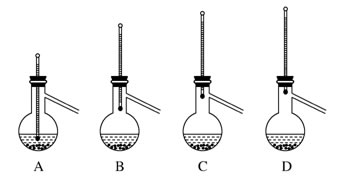
| C�����Ҵ���ˮ���룬��ͼ1��ͼ2���� |
| D��ϡ��ŨH2SO4����ͼ3���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
[���]ÿƬ������������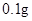 [��Ӧ֢]����ȱ����ƶѪ֢��Ԥ���������á� [�����÷�]����Ԥ���� 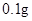 /�գ����������� /�գ�����������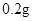 �� ��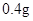 /�ա� /�ա�С������Ԥ���� 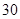 �� ��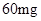 /�գ������� /�գ�������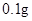 �� ��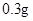 /�� /��[����]�ܹ⡢�ܷ⡢�ڸ��ﴦ���档 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�����Թ�ȡ���Լ�ƿ�е�NaOH��Һ������ȡ�����࣬Ϊ�˲��˷ѣ��ְѹ������Լ������Լ�ƿ�� |
| B��������Ӧʵ���ķ�Һ���ȵ���ˮ���У�����ˮ������ˮ�� |
| C�����Թ��еμ�Һ��ʱ����ͷ�ιܱ�������Թ��ڱڣ�����Һ�彦�� |
| D����Ũ��������һ�����ʵ���Ũ�ȵ�ϡ����ʱ��Ũ��������ˮ��Ӧ��ȴ�����²���ת�Ƶ�����ƿ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
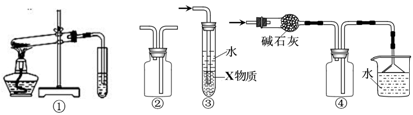
| A��װ�âٿ�����̽��NH4HCO3�����ȶ��� |
| B��װ�âڿ������ռ�H2��CO2 |
| C��װ�â���X��ΪCCl4�����������հ������Ȼ��⣬���ܷ����� |
| D��װ�âܿ����ڸ���ռ�NH3�������ն���İ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 12Na2CrO4+3Fe2O3+7KCl+12H2O��
12Na2CrO4+3Fe2O3+7KCl+12H2O��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com