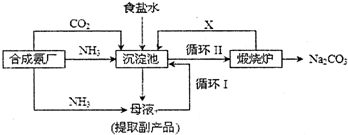
��ҵ�϶Ժ�ˮ��Դ�ۺϿ������õIJ��ֹ���������ͼ1��ʾ��
��1���������Ca
2+��Mg
2+��SO
42-�����ʣ����ƺ�ɵñ���NaCl��Һ������ʱͨ������Һ�������м��������BaCl
2��Һ��������NaOH��Һ������Na
2CO
3��Һ�����˺�����Һ�м�����������Һ�����ԣ���д������Na
2CO
3��Һ����ػ�ѧ��Ӧ�����ӷ���ʽ��
Ba2++CO32-=BaCO3����Ca2++CO32-=CaCO3����
Ba2++CO32-=BaCO3����Ca2++CO32-=CaCO3����
��
��2���������������Ⱥ��Ƶ�Br
2��CaSO
4��Mg��OH��
2���ܷ�Br
2��Mg��OH��
2��CaSO
4��˳���Ʊ���
��
��
����ܡ�����ԭ����
����ȳ���Mg��OH��2��������л������CaSO4��������Ʒ����
����ȳ���Mg��OH��2��������л������CaSO4��������Ʒ����
��
��3���Ȼ�������Ҫ���ȼҵ������ԭ�ϣ���ⱥ��ʳ��ˮ��������Ĥ���ۺ�Ĥ���ۣ�����Ĥ��Ĥ������ͨ���ķ��ӻ�������
B
B
��
A��Cl
- B��Na
+ C��OH
- D��Cl
2��4������ʳ��ˮ���ʱ���Դ���������ĵ缫�Ϸ����ķ�ӦΪ
������Ӧ��2Cl--2e-=Cl2��
������Ӧ��2Cl--2e-=Cl2��
�����Դ���������ĵ缫������ҺpH
���
���
������䡢��С����
��5���Ȼ��ƵĿ�������������ҹ���ѧ�Һ�°�ĸ�����������գ��������̼�Ҫ��ʾ��ͼ2��
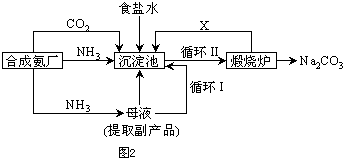
��������������ķ�����
�����Ƽ
�����Ƽ
������Ʒ��һ����;Ϊ
������
������
��
д������������X���ʵķ���ʽ
CO2
CO2
��ʹԭ���Ȼ��Ƶ������ʴ�70%��ߵ�90%���ϣ���Ҫ�������
��
��
�������������еı�ţ���ѭ�����ӳ�������ȡ�������IJ�����
����
����
��
�ںϳɰ�ԭ�����е����Ʊ��ķ���֮һΪ
Һ̬�������ݷе����
Һ̬�������ݷе����
����һԭ������������ȡ��ѧ����ʽΪ
��
�۳������з����Ļ�ѧ��Ӧ����ʽ��
NH3+CO2+NaCl+H2O=NH4Cl+NaHCO3��
NH3+CO2+NaCl+H2O=NH4Cl+NaHCO3��
��Ҫʵ�ָ÷�Ӧ������ΪӦ����β�����
���ı����Ȼ�����Һ��ͨ�������̼����õ�̼�����ƾ���
���ı����Ȼ�����Һ��ͨ�������̼����õ�̼�����ƾ���
��
��Ϊ�����Ʒ̼�������Ƿ����Ȼ��ƣ���ȡ������������ˮ���ٵμ�
�������ữ�����������۲������ɫ����
�������ữ�����������۲������ɫ����
��





 ���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д�
���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д� ����ѵ��ϵ�д�
����ѵ��ϵ�д�