
·Ö±šÓÉC”¢H”¢OČżÖÖŌŖĖŲ×é³ÉµÄÓŠ»śĪļA”¢B”¢C»„ĪŖĶ¬·ÖŅģ¹¹Ģ壬ĖüĆĒ·Ö×ÓÖŠC”¢H”¢OŌŖĖŲµÄÖŹĮæ±ČĪŖ15©U2©U8£¬ĘäÖŠ»ÆŗĻĪļAµÄÖŹĘ×Ķ¼ČēĻĀ”£
AŹĒÖ±Į“½į¹¹£¬ĘäŗĖ“Ź²ÕńĒāĘ×ÓŠČż×é·å£¬ĒŅ·åĆ껿֮±ČĪŖ1©U1©U2£¬ĖüÄܹ»·¢ÉśŅų¾µ·“Ó¦”£BĪŖĪåŌŖ»·õ„”£CµÄŗģĶā¹āĘ×±ķĆ÷Ęä·Ö×ÓÖŠ“ęŌŚ¼×»ł”£ĘäĖüĪļÖŹµÄ×Ŗ»Æ¹ŲĻµČēĻĀ£ŗ
£Ø1£©IµÄ·Ö×ÓŹ½ŹĒ£ŗ ”£I·Ö×ÓÖŠµÄ¹ŁÄÜĶÅĆū³ĘŹĒ£ŗ ”£
£Ø2£©DŗĶHµÄ½į¹¹¼ņŹ½·Ö±šŹĒ£ŗ ”¢ ”£
£Ø3£©Š“³öĻĀĮŠ·“Ó¦·½³ĢŹ½(ÓŠ»śĪļÓĆ½į¹¹¼ņŹ½±ķŹ¾)
D”śC £»
H”śG ”£
£Ø8·Ö£©£Ø1£©C5H8O4£»ōČ»ł £Ø2£©CH3CH£ØOH£©CH2CH2COOH£¬OHCCH2CH2CH2COOH
£Ø3£© D”śC£ŗHOOCCH2CH2CH(OH)CH3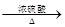 HOOCCH2CH£½CHCH3+H2O£»
HOOCCH2CH£½CHCH3+H2O£»
H”śG£ŗHOOCCH2CH2CH2CHO+H2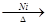 HOOCCH2CH2CH2CH2OH
HOOCCH2CH2CH2CH2OH
½āĪöŹŌĢā·ÖĪö£ŗ·Ö×ÓÖŠC”¢H”¢OŌŖĖŲµÄÖŹĮæ±ČĪŖ15©U2©U8£¬Ōņ·Ö×ÓÖŠC”¢H”¢OŌŖĖŲŌ×ÓµÄøöŹżÖ®±ČŹĒ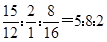 ”£øł¾ŻAµÄÖŹĘ×Ķ¼æÉÖŖ£¬AµÄĻą¶Ō·Ö×ÓÖŹĮæŹĒ100£¬ĖłŅŌAµÄ·Ö×ÓŹ½ŹĒC5H802”£AŹĒÖ±Į“½į¹¹£¬ĘäŗĖ“Ź²ÕńĒāĘ×ÓŠČż×é·å£¬ĒŅ·åĆ껿֮±ČĪŖ1©U1©U2£¬ĖüÄܹ»·¢ÉśŅų¾µ·“Ó¦£¬ĖµĆ÷ŗ¬ÓŠČ©»ł£¬Ņņ“ĖAµÄ½į¹¹¼ņŹ½ŹĒOHCCH2CH2CH2CHO”£A·¢ÉśŅų¾µ·“Ӧɜ³ÉI£¬ŌņIµÄ½į¹¹¼ņŹ½ŹĒHOOCCH2CH2CH2COOH”£BĪŖĪåŌŖ»·õ„£¬ŌŚĒāŃõ»ÆÄĘČÜŅŗÖŠĖ®½āÉś³ÉD”£DŌŚÅØĮņĖįµÄ×÷ÓĆĻĀ·¢ÉśĻūČ„·“Ӧɜ³ÉC£¬CµÄŗģĶā¹āĘ×±ķĆ÷Ęä·Ö×ÓÖŠ“ęŌŚ¼×»ł£¬ĒŅC×īÖÕ×Ŗ»ÆĪŖI£¬ĖłŅŌDµÄ½į¹¹¼ņŹ½ŹĒCH3CH£ØOH£©CH2CH2COOH£¬ŌņBµÄ½į¹¹¼ņŹ½ŹĒ
”£øł¾ŻAµÄÖŹĘ×Ķ¼æÉÖŖ£¬AµÄĻą¶Ō·Ö×ÓÖŹĮæŹĒ100£¬ĖłŅŌAµÄ·Ö×ÓŹ½ŹĒC5H802”£AŹĒÖ±Į“½į¹¹£¬ĘäŗĖ“Ź²ÕńĒāĘ×ÓŠČż×é·å£¬ĒŅ·åĆ껿֮±ČĪŖ1©U1©U2£¬ĖüÄܹ»·¢ÉśŅų¾µ·“Ó¦£¬ĖµĆ÷ŗ¬ÓŠČ©»ł£¬Ņņ“ĖAµÄ½į¹¹¼ņŹ½ŹĒOHCCH2CH2CH2CHO”£A·¢ÉśŅų¾µ·“Ӧɜ³ÉI£¬ŌņIµÄ½į¹¹¼ņŹ½ŹĒHOOCCH2CH2CH2COOH”£BĪŖĪåŌŖ»·õ„£¬ŌŚĒāŃõ»ÆÄĘČÜŅŗÖŠĖ®½āÉś³ÉD”£DŌŚÅØĮņĖįµÄ×÷ÓĆĻĀ·¢ÉśĻūČ„·“Ӧɜ³ÉC£¬CµÄŗģĶā¹āĘ×±ķĆ÷Ęä·Ö×ÓÖŠ“ęŌŚ¼×»ł£¬ĒŅC×īÖÕ×Ŗ»ÆĪŖI£¬ĖłŅŌDµÄ½į¹¹¼ņŹ½ŹĒCH3CH£ØOH£©CH2CH2COOH£¬ŌņBµÄ½į¹¹¼ņŹ½ŹĒ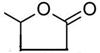 ”£ŌņCµÄ½į¹¹¼ņŹ½ŹĒHOOCCH2CH£½CHCH3”£EæÉŅŌĖ®½ā·“Ó¦£¬ĖµĆ÷EŹĒĀ±“śĢž”£EĖ®½āÉś³ÉF£¬¶ųFÓÖæÉŅŌ·¢Éś¼Ó³É·“Ó¦£¬ĖµĆ÷FÖŠ»¹ŗ¬ÓŠĢ¼Ģ¼Ė«¼ü£¬ŌņEµÄ½į¹¹¼ņŹ½ŹĒClCH2CH£½CHCH2COOH£¬FµÄ½į¹¹¼ņŹ½ŹĒHOCH2CH£½CHCH2COOH”£GµÄ½į¹¹¼ņŹ½ŹĒHOOCCH2CH2CH2CH2OH”£G·¢Éś“ß»ÆŃõ»ÆÉś³ÉH£¬ĖłŅŌHµÄ½į¹¹¼ņŹ½ŹĒHOOCCH2CH2CH2CHO£¬H¼ĢŠų·¢ÉśŃõ»Æ·“Ӧɜ³ÉI”£
”£ŌņCµÄ½į¹¹¼ņŹ½ŹĒHOOCCH2CH£½CHCH3”£EæÉŅŌĖ®½ā·“Ó¦£¬ĖµĆ÷EŹĒĀ±“śĢž”£EĖ®½āÉś³ÉF£¬¶ųFÓÖæÉŅŌ·¢Éś¼Ó³É·“Ó¦£¬ĖµĆ÷FÖŠ»¹ŗ¬ÓŠĢ¼Ģ¼Ė«¼ü£¬ŌņEµÄ½į¹¹¼ņŹ½ŹĒClCH2CH£½CHCH2COOH£¬FµÄ½į¹¹¼ņŹ½ŹĒHOCH2CH£½CHCH2COOH”£GµÄ½į¹¹¼ņŹ½ŹĒHOOCCH2CH2CH2CH2OH”£G·¢Éś“ß»ÆŃõ»ÆÉś³ÉH£¬ĖłŅŌHµÄ½į¹¹¼ņŹ½ŹĒHOOCCH2CH2CH2CHO£¬H¼ĢŠų·¢ÉśŃõ»Æ·“Ӧɜ³ÉI”£
æ¼µć£ŗæ¼²éÓŠ»śĪļ»ÆѧŹ½”¢¹ŁÄÜĶÅ”¢½į¹¹¼ņŹ½µÄÅŠ¶ĻŅŌ¼°·½³ĢŹ½µÄŹéŠ“µČ
µćĘĄ£ŗøĆĢāŹĒøßæ¼ÖŠµÄ³£¼ūĢāŠĶ£¬ŹōÓŚÖŠµČÄѶȵďŌĢā”£ŹŌĢāĢł½üøßæ¼£¬×ŪŗĻŠŌĒ棬ŌŚ×¢ÖŲ¶Ōѧɜ»ł“”ÖŖŹ¶¹®¹ĢÓėѵĮ·µÄĶ¬Ź±£¬²ąÖŲ¶ŌѧɜÄÜĮ¦µÄÅąŃųÓė½āĢā·½·ØµÄÖøµ¼ŗĶѵĮ·”£øĆĄąŹŌĢāÄܽĻČ«ĆęµŲæ¼²éѧɜµÄÓŠ»ś»Æѧ»ł“”ÖŖŹ¶ŗĶĀß¼Ė¼Ī¬ÄÜĮ¦”¢““ŌģĖ¼Ī¬ÄÜĮ¦£¬ĢįøßѧɜµÄÓ¦ŹŌÄÜĮ¦ŗĶ“šĢāŠ§ĀŹ£¬Ņ²ÓŠĄūÓŚÅąŃųѧɜµÄ×ŌѧÄÜĮ¦ŗĶÖŖŹ¶µÄĒØŅĘÄÜĮ¦”£øĆĢāµÄ¹Ų¼üŹĒ¼Ē×”³£¼ū¹ŁÄÜĶŵĽį¹¹”¢ŠŌÖŹŅŌ¼°¹ŁÄÜĶÅÖ®¼äµÄĻą»„×Ŗ»Æ£¬Č»ŗó½įŗĻĢāŅāĮé»īŌĖÓĆ¼“æÉ”£



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĶʶĻĢā
ŅŃÖŖÓŠ»śĪļA”«IÖ®¼äµÄ×Ŗ»Æ¹ŲĻµČēĶ¼24ĖłŹ¾£ŗ
¢ŁAÓėD”¢BÓėE”¢IÓėF»„ĪŖĶ¬·ÖŅģ¹¹Ģå
¢Ś¼ÓČČĢõ¼žĻĀŠĀÖĘCu(OH)2Šü×ĒŅŗ·Ö±š¼ÓČėµ½ÓŠ»śĪļI”¢FÖŠ£¬IÖŠĪŽĆ÷ĻŌĻÖĻó£¬FÖŠ±äשŗģÉ«
¢ŪCµÄ×ī¼ņŹ½ÓėŅŅČ²ĻąĶ¬£¬ĒŅĻą¶Ō·Ö×ÓÖŹĮæĪŖ104
¢ÜBŹĒŅ»ÖÖĶ¬·ÖŅģ¹¹ĢåÓėFeC13·¢ÉśĻŌÉ«·“Ó¦”£
øł¾ŻŅŌÉĻŠÅĻ¢£¬»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©CÖŠŗ¬ÓŠµÄ¹ŁÄÜĶÅĆū³ĘĪŖ ”£
£Ø2£©HµÄ½į¹¹¼ņŹ½ĪŖ ”£
£Ø3£©·“Ó¦¢Ł”«¢įÖŠŹōÓŚČ”“ś·“Ó¦µÄŹĒ ”£
£Ø4£©Š“³öFÓėŠĀÖĘCu(OH)2Šü×ĒŅŗ·“Ó¦µÄ»Æѧ·½³ĢŹ½ ”£
£Ø5£©±½»·ÉĻŗ¬ÓŠĮ½øöČ”“ś»łĒŅÄÜÓėNaOHČÜŅŗ·“Ó¦£¬µ«²»ÓėFeC13·¢ÉśĻŌÉ«·“Ó¦µÄGµÄĶ¬·ÖŅģ¹¹ĢåÓŠ ÖÖ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĶʶĻĢā
 ŹĒŅ»ÖÖŅ½Ņ©ÖŠ¼äĢ壬³£ÓĆĄ“Öʱøæ¹ÄżŃŖŅ©£¬æÉĶعżĻĀĮŠĀ·ĻßŗĻ³É
ŹĒŅ»ÖÖŅ½Ņ©ÖŠ¼äĢ壬³£ÓĆĄ“Öʱøæ¹ÄżŃŖŅ©£¬æÉĶعżĻĀĮŠĀ·ĻßŗĻ³É
ŅŃÖŖ£ŗE±ä³ÉFĻąµ±ÓŚŌŚE·Ö×ÓÖŠČ„µō1øöX·Ö×Ó”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©A”śBµÄ·“Ó¦ĄąŠĶŹĒ ;
£Ø2£©E·Ö×ÓÖŠÓŠ ÖÖ²»Ķ¬»Æѧ»·¾³µÄĒāŌ×Ó£»
£Ø3£©X“ß»ÆŃõ»ÆæÉÉś³ÉŅ»ÖÖ³£ĪĀĻĀĪŖĘųĢåµÄĪļÖŹ£¬Š“³öøĆĘųĢå·Ö×ӵĽį¹¹Ź½ £»
£Ø4£©Š“³öD”śEµÄ»Æѧ·½³ĢŹ½ £»
£Ø5£©ŗ¬õ„»łĒŅÓöFeCl3ČÜŅŗĻŌ×ĻÉ«£¬ÓėC»„ĪŖĶ¬·ÖŅģ¹¹ĢåµÄÓŠ»śĪļÓŠ ÖÖ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĶʶĻĢā
ĄūµØ½ā¾·Ņ©”°ĮĮ¾ś¼×ĖŲ”±øś(CH3)2SO4·“Ó¦æÉŅŌÉś³ÉA£¬¶žÕߵĽį¹¹¼ņŹ½ČēĻĀĶ¼”£
ŹŌ»Ų“šĻĀĮŠÓŠ¹ŲĪŹĢā£ŗ
£Ø1£©”°ĮĮ¾ś¼×ĖŲ”±µÄ·Ö×ÓŹ½ĪŖ_______£»1 mol”°ĮĮ¾ś¼×ĖŲ”±øśÅØäåĖ®·“Ó¦Ź±×ī¶ąĻūŗÄ_____ mol Br2”£
£Ø2£©ÓŠ»śĪļAÄÜ·¢ÉśČēĻĀ×Ŗ»Æ”£ĘäÖŠG·Ö×ÓÖŠµÄĢ¼Ō×ÓŌŚŅ»ĢõÖ±ĻßÉĻ”£
ŅŃÖŖ£ŗ¢Å
¢ĘR?O?CH3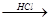 R?OH£ØRĪŖÖ¬·¾Ģž»ņ·¼ĻćĢžµÄĢž»ł£©
R?OH£ØRĪŖÖ¬·¾Ģž»ņ·¼ĻćĢžµÄĢž»ł£©
¢ŁC”śDµÄ»Æѧ·½³ĢŹ½ŹĒ_________________________________________________£»
¢ŚGµÄ½į¹¹¼ņŹ½ŹĒ___ £»
¢ŪĶ¬Ź±·ūŗĻĻĀĮŠĢõ¼žµÄEµÄĶ¬·ÖŅģ¹¹ĢåÓŠ ÖÖ£»
a£®ÓöFeCl3ČÜŅŗĻŌÉ«£»
b£®²»ÄÜ·¢ÉśĖ®½ā·“Ó¦£¬ÄÜ·¢ÉśŅų¾µ·“Ó¦£»
c£®±½»·ÉĻµÄŅ»ĀČČ”“śĪļÖ»ÓŠŅ»ÖÖ£¬·Ö×ÓÖŠĪŽ¼×»ł
Š“³öĘäÖŠÖĮÉŁ2ÖֵĽį¹¹¼ņŹ½£ŗ___________________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĶʶĻĢā
ŅŌŅŅČ²ĪŖŌĮĻĶعżŅŌĻĀĮ÷³ĢÄÜŗĻ³ÉÓŠ»śĪļÖŠ¼äĢåD”£
ŅŃÖŖ£ŗ£Ø1£©
£Ø2£©
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©»ÆŗĻĪļDµÄ·Ö×ÓŹ½ĪŖ £¬Š“³öDÖŠŅ»ÖÖ¹ŁÄÜĶŵÄĆū³Ę ”£
£Ø2£©Š“³öÉś³ÉAµÄ»Æѧ·“Ó¦·½³ĢŹ½£ŗ ”£
£Ø3£©»ÆŗĻĪļBŌŚÅØĮņĖį“ß»ÆĻĀ£¬¼ÓČČÓėHOOCCOOH·“Ӧɜ³É»·×“õ„µÄ»Æѧ·½³ĢŹ½ĪŖ£ŗ
(×¢Ć÷Ģõ¼ž)”£
£Ø4£©»ÆŗĻĪļCµÄ½į¹¹¼ņŹ½ĪŖ£ŗ ”£
£Ø5£©ŅŃÖŖ1mol HCHOŗĶ1mol CH3CH2CHO·¢ÉśĄąĖĘŅŃÖŖ£Ø2£©µÄ·“Ó¦£¬Éś³É1molE”£ŅŌĻĀ¹ŲÓŚEµÄĖµ·ØÕżČ·µÄŹĒ ”£
a”¢EÄÜÓėH2·¢Éś¼Ó³É·“Ó¦£¬Ņ²ÄÜŹ¹ĖįŠŌøßĆĢĖį¼ŲČÜŅŗĶŹÉ«
b”¢EŹōÓŚõ„ĄąĪļÖŹ
c”¢1molEĶźČ«Č¼ÉÕŠčĻūŗÄ5molO2
d”¢Éś³ÉEµÄ·“Ó¦ŹōÓŚČ”“ś·“Ó¦
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĶʶĻĢā
²¼f²¼Āå·ŅŹĒŅ»ÖÖĻūŃ×ÕņĶ“µÄŅ©Īļ”£ĖüµÄ¹¤ŅµŗĻ³ÉĀ·ĻßČēĻĀ£ŗ
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©»ÆŗĻĪļAµÄ·Ö×ÓŹ½ĪŖ ”£
£Ø2£©A³¤ĘŚ±©Ā¶ŌŚæÕĘųÖŠ»į±äÖŹ£¬ĘäŌŅņŹĒ ”£
£Ø3£©ÓÉAµ½BµÄ·“Ó¦Ķس£ŌŚµĶĪĀŹ±½ųŠŠ”£ĪĀ¶ČÉżøߏ±£¬¶ąĻõ»łČ”“śø±²śĪļ»įŌö¶ą”£ĻĀĮŠ¶žĻõ»łČ”“śĪļÖŠ£¬×īæÉÄÜÉś³ÉµÄŹĒ (Ģī×ÖÄø£¬2·Ö)”£
a£® b£®
b£® c£®
c£® d£®
d£®
£Ø4£©B±»H2»¹ŌµÄ²śĪļCµÄ½į¹¹¼ņŹ½ĪŖ ”£
£Ø5£©D”śEµÄ»Æѧ·½³ĢŹ½ĪŖ£ŗ ”£
£Ø6£©FµÄ½į¹¹¼ņŹ½ ”£
£Ø7£©DµÄĶ¬·ÖŅģ¹¹ĢåHŹĒŅ»ÖÖ¦Į-°±»łĖį£¬Hæɱ»ĖįŠŌKMnO4ČÜŅŗŃõ»Æ³É¶Ō±½¶ž¼×Ėį£¬ŌņHµÄ½į¹¹¼ņŹ½ŹĒ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĶʶĻĢā
(12·Ö)ĪŅ¹ś±ōĮŁŗ£ÓņµÄŹ”·Ż£¬Äܹ»³ä·ÖĄūÓĆŗ£Ė®ÖʵƶąÖֻƹ¤²śĘ·”£ČēĶ¼ŹĒŅŌŗ£Ė®”¢±“æĒ(Ö÷ŅŖ³É·ÖCaCO3)µČĪŖŌĮĻÖĘČ”¼øÖֻƹ¤²śĘ·µÄ¹¤ŅÕĮ÷³ĢĶ¼£¬ĘäÖŠ¹ĢĢåBŹĒÉś»īÖŠµÄŅ»ÖÖ±ŲŠčĘ·£¬EŹĒŅ»ÖֻƷŹ£¬NŹĒŅ»ÖÖ³£¼ūµÄ½šŹōµ„ÖŹ”£
½įŗĻÉĻŹö¹¤ŅÕĮ÷³Ģ»Ų“šĻĀĮŠĪŹĢā”£
£Ø1£©ĪļÖŹHŗĶLµÄ»ÆѧŹ½·Ö±šĪŖ ŗĶ ”£
£Ø2£©ÉĻŹö¹¤ŅÕĮ÷³ĢÖŠæÉŅŌŃ»·Ź¹ÓƵÄĪļÖŹµÄ»ÆѧŹ½ĪŖ____________”£
£Ø3£©·“Ó¦¢ŁµÄ»Æѧ·½³ĢŹ½ĪŖ £»ŌŚ·“Ó¦¢ŁÖŠ±ŲŠėĻČĶØČėNH3£¬¶ųŗóĶØČėD£¬ĘäŌŅņŹĒ ”£
£Ø4£©¹¤ŅµÉĻĄūÓĆFæÉÖʵĆĮķŅ»Öֻƹ¤²śĘ·£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø5£©ÓÉKČÜŅŗ¾ČēŗĪ“¦Ąķ²ÅÄÜÖʵĆN£æ¹¤ŅµÉĻ½«KČÜŅŗŌŚ µÄĢõ¼žÕō·¢½į¾§ÖʵĆĪŽĖ®ŃĪ£¬Č»ŗóŌŁµē½āĘäČŪČŚŃĪ£¬Š“³öµē½āµÄ»Æѧ·“Ó¦·½³ĢŹ½ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014-2015ѧğøŹĖąŹ”ø߶žÉĻµŚŅ»“ĪŌĀæ¼»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
¼×ĶéŹĒŅ»ÖÖøߊ§Ēå½ąµÄŠĀÄÜŌ“£¬0£®25mol¼×ĶéĶźČ«Č¼ÉÕÉś³ÉŅŗĢ¬Ė®Ź±·Å³ö222£®5KJČČĮ棬ŌņĻĀĮŠČČ»Æѧ·½³ĢŹ½ÖŠÕżČ·µÄŹĒ( )
A£®2CH4(g) + 4O2(g) == 2CO2(g) + 4H2O(l)£»¦¤H== +890KJ”¤mol”Ŗ1
B£® CH4(g) + 2O2(g) == CO2(g) +2H2O(l)£»¦¤H== +890KJ”¤mol”Ŗ1
C£® CH4(g) + 2O2(g) == CO2(g) +2H2O(l)£»¦¤H== -890KJ”¤mol”Ŗ1
D£® 2CH4(g) + 4O2(g) == 2CO2(g) + 4H2O(l)£»¦¤H== -890KJ”¤mol”Ŗ1
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014-2015ѧğøŹĖąŹ”ø߶žµŚŅ»Ń§ĘŚĘŚÖŠæ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
·“Ó¦NH4HS(s)  NH3(g)+H2S(g),ŌŚÄ³Ņ»ĪĀ¶ČĻĀ“ļµ½Ę½ŗā”£ĻĀĮŠø÷ÖÖĒéæöÖŠ,²»ÄÜŹ¹»ÆŃ§Ę½ŗā·¢ÉśŅĘ¶ÆµÄŹĒ
NH3(g)+H2S(g),ŌŚÄ³Ņ»ĪĀ¶ČĻĀ“ļµ½Ę½ŗā”£ĻĀĮŠø÷ÖÖĒéæöÖŠ,²»ÄÜŹ¹»ÆŃ§Ę½ŗā·¢ÉśŅĘ¶ÆµÄŹĒ
A£®ĘäĖūĢõ¼ž²»±äŹ±ĶØČėSO2ĘųĢå B£®ŅĘ×ßŅ»²æ·ÖNH4HS¹ĢĢå
C£®ČŻĘ÷Ģå»ż²»±ä,³äČėNH3 D£®Ń¹Ēæ²»±äŹ±,³äČėN2
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com