
NiSO4•6H2O��һ����ɫ������ˮ�ľ��壬�㷺���ڻ�ѧ������������صȣ����ɵ�Ʒ������������⣬�����У�Cu��Zn��Fe��Cr��Ԫ�صĻ��������ʣ�Ϊԭ�ϻ�á��йع����������£�

��1���������м�H2SO4������Ҫ��ֽ��裬��Ŀ���� ��
��2����Na2S��Ŀ���dz�ȥͭ��п�����ʣ���д����ȥCu2+�����ӷ���ʽ ��
��3����6%��H2O2ʱ���¶Ȳ��ܹ��ߣ���Ŀ���� ��
��4��������������H2O2�������������NaOH����pHֵ2��4��Χ�������������������������������У�����������NaClO3���棬��NaClO3����Fe2+�����ӷ���ʽΪ ��
��5��������������ҺIII�����ʵ���Ҫ�ɷ��ǣ� ��
��6������I�������¹��̣����ˣ���H2SO4�ܽ⣬ �� �����ˡ�ϴ�ӻ�ò�Ʒ��
��16�֣���1���ӿ췴Ӧ���ʻ���߽����ʣ������ĸ��֣� ��2�֣�
��2��S2��+Cu2+=CuS����2�֣�
��3�����٣���ֹ����������ķֽ� ��2�֣�
��4��6Fe2++ClO3��+6H+=6Fe3++Cl��+3H2O��2�֣�
��5��Na2SO4��NiSO4 ��4�֣�©ѡ��2�֣���ѡ�����֣�
��6������Ũ�� ��ȴ�ᾧ��4�֣�
��������
�����������1�����ݻ�ѧ��Ӧ���ʵ�Ӱ�����ؼ����ɿ�֪����ֽ����Ŀ���Ǽӿ�����е������ʵ������ᷴӦ�����ʣ������Ԫ�صĽ����ʣ���2��Na2S�ǿ������Σ�����ˮʱ�����S2����S2����Cu2+��Ӧ����CuS��������3��������������ɫ����������Ŀ���ǽ�Fe2+��ȫ����ΪFe3+���Ҳ������µ����ʣ�����H2O2���ȶ��������ֽ�Ϊˮ������������¶Ȳ��ܹ��ߣ���4��NaClO3�ǿ������Σ�NaOH��ǿ����������֪��Fe2+�ǻ�ԭ����ClO3������������Fe3+�����������ԭ������Cl����������Cl2����Ϊ������Һ���ܴ�������Cl2��������Ԫ�ػ��ϼ���������������Ԫ�ػ��ϼ۽�������������غ㡢ԭ���غ�ԭ���ɵ÷�Ӧʽ��6Fe2++ClO3��+6H+=6Fe3++Cl��+3H2O����5����ͼ��֪����Na2S��Ŀ���dz�ȥͭ��п�����ʣ���H2O2��NaOH��Ŀ���dz�ȥ�������ʣ���Na2CO3��Ŀ���dz�ȥ�������ʣ�����ƶ���Һ1����Ҫ�ɷ���NiSO4��FeSO4��Cr2(SO4)3��Na2SO4����ҺII����Ҫ�ɷ���NiSO4��Cr2(SO4)3��Na2SO4����ҺIII����Ҫ�ɷ���NiSO4��Na2SO4����6����NiSO4��Na2SO4������Ļ����Һ����ȡNiSO4•6H2O���壬��Ҫ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����ȹ��̡�
���㣺���������Ʊ��������̴��⣬�漰���Ͳ�����Ŀ�ġ�����ʵ�ʡ��¶ȿ��Ƶ�ԭ����Һ��Ҫ�ɷ֡������Ʊ��Ļ���������֪ʶ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
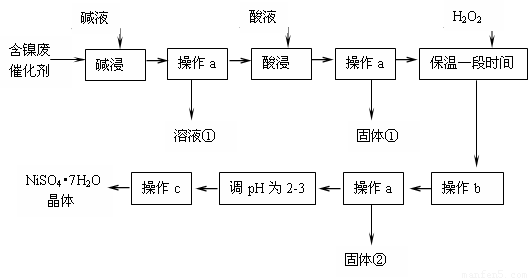
��14�֣�ij��ѧ�о���ѧϰС��ͨ���������ϣ����������ͼ��ʾ�ķ����Ժ����ϴ���Ϊԭ�����Ʊ�NiSO4•7H2O����֪ij�������ĺ���������Ҫ����Ni��������Al��31%����Fe��1.3%���ĵ��ʼ������������������(3.3%)��
����������������������ʽ��ȫ����ʱ��pH���£�
| ������ | Al(OH)3 | Fe(OH)3 | Fe(OH)2 | Ni(OH)2 |
| pH | 5.2 | 3.2 | 9.7 | 9.2 |
�ش��������⣺
�� ����a��c����ʹ�õ�����������̨������Ȧ�����ƾ��ơ��ձ������������Ҫ����Ҫ����Ϊ ��
�� ������������з��������ӷ���ʽ�� ��
�ǡ������ʱ����������� (�ѧʽ�������������a���������ٺ���Һ�п��ܺ��еĽ��������� ��
�� ����bΪ������Һ��pH������ΪpH����ѵ��ط�Χ�� ��
�ɡ���pHΪ2~3����Ŀ���� ��
�� ��Ʒ��������ʱ����������̷���FeSO4��7H2O������ԭ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�꽭��ʡ�߶���ѧ����ĩ���Ի�ѧ ���ͣ�ʵ����
��14�֣�ij��ѧ�о���ѧϰС��ͨ���������ϣ����������ͼ��ʾ�ķ����Ժ����ϴ���Ϊԭ�����Ʊ�NiSO4•7H2O����֪ij�������ĺ���������Ҫ����Ni��������Al��31%����Fe��1.3%���ĵ��ʼ������������������(3.3%)��
����������������������ʽ��ȫ����ʱ��pH���£�
|
������ |
Al(OH)3 |
Fe(OH)3 |
Fe(OH)2 |
Ni(OH)2 |
|
pH |
5.2 |
3.2 |
9.7 |
9.2 |
�ش��������⣺
�� ����a��c����ʹ�õ�����������̨������Ȧ�����ƾ��ơ��ձ������������Ҫ����Ҫ����Ϊ ��
�� ������������з��������ӷ���ʽ�� ��
�ǡ������ʱ����������� (�ѧʽ�������������a���������ٺ���Һ�п��ܺ��еĽ��������� ��
�� ����bΪ������Һ��pH������ΪpH����ѵ��ط�Χ�� ��
�ɡ���pHΪ2~3����Ŀ���� ��
�� ��Ʒ��������ʱ����������̷���FeSO4��7H2O������ԭ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��14�֣�ij��ѧ�о���ѧϰС��ͨ���������ϣ����������ͼ��ʾ�ķ����Ժ����ϴ���Ϊԭ�����Ʊ�NiSO4•7H2O����֪ij�������ĺ���������Ҫ����Ni��������Al��31%����Fe��1.3%���ĵ��ʼ������������������(3.3%)��

����������������������ʽ��ȫ����ʱ��pH���£�
| ������ | Al(OH)3 | Fe(OH)3 | Fe(OH)2 | Ni(OH)2 |
| pH | 5.2 | 3.2 | 9.7 | 9.2 |
�ش��������⣺
�� ����a��c����ʹ�õ�����������̨������Ȧ�����ƾ��ơ��ձ������������Ҫ����Ҫ����Ϊ ��
�� ������������з��������ӷ���ʽ�� ��
�ǡ������ʱ����������� (�ѧʽ�������������a���������ٺ���Һ�п��ܺ��еĽ��������� ��
�� ����bΪ������Һ��pH������ΪpH����ѵ��ط�Χ�� ��
�ɡ���pHΪ2~3����Ŀ���� ��
�� ��Ʒ��������ʱ����������̷���FeSO4��7H2O������ԭ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij��ѧ�о���ѧϰС��ͨ���������ϣ����������ͼ��ʾ�ķ����Ժ����ϴ���Ϊԭ�����Ʊ�NiSO4•7H2O����֪ij�������ĺ���������Ҫ����Ni��������Al��31%����Fe��1.3%���ĵ��ʼ������������������(3.3%)��
 |
����������������������ʽ����ʱ��pH���£�
| ������ | ��ʼ���� | ��ȫ���� |
| Al(OH)3 | 3.8 | 5.2 |
| Fe(OH)3 | 2.7 | 3.2 |
| Fe(OH)2 | 7.6 | 9.7 |
| Ni(OH)2 | 7.1 | 9.2 |
�ش��������⣺
��1������a��c����ʹ�õ�����������̨������Ȧ�����ƾ��ơ��ձ������������Ҫ����Ҫ����Ϊ �� ��
��2��������������з��������ӷ���ʽ�� �� �� �� ��
��3���������ʱ����������� �� ���ѧʽ�������������a���������ٺ���Һ�п��ܺ��еĽ��������� �� ��
��4�� ����bΪ������Һ��pH������ΪpH����ѵ��ط�Χ�� �� ��
��5������pHΪ2~3����Ŀ���� �� ��
��6����Ʒ��������ʱ����������̷���FeSO4��7H2O������ԭ������� �� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com