

| A��С�ձ��ڲ�����ˮ����ʹ��õķ�Ӧ����ֵƫС |
| B���ᡢ����ʱ��Ӧ����Ͳ�е���Һ���������ձ�����Һ�У��Է�Һ���⽦ |
| C���ձ���������ֽ���������ǹ̶�С�ձ� |
| D��������ͬŨ�Ⱥ�������������ϡ������Һ����ʵ�� |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
| 1 |
| 2 |
| 1 |
| 2 |
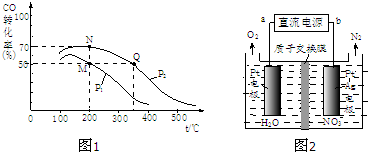
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��CH3COONa��Һ�м�������KNO3�����ļ�����Һһ���У� C(Na+)��c(CH3COO��)=c(OH��)��c(H+) |
| B����ZnS��ɫ����Һ�м���CuSO4��Һ���к�ɫ��������������ΪKsp (ZnS)��Ksp (CuS) |
| C������ڵ�NaOH��Һ�еμ�FeCl3������Һ�������Ʊ�Fe(OH)3���� |
| D����ˮ�м���NaCl�ܽ������ͺ���Һ��pH����7 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��Vһ������0.2 |
| B��V���ڻ����0.2 |
C�������Һ������Ũ��һ�����㣺c(Cl��)>c(NH )> c(H��)>c(OH��) )> c(H��)>c(OH��) |
D�������Һ������Ũ��һ�����㣺c(NH )��c(H��)�� c(Cl��)��c(OH��) )��c(H��)�� c(Cl��)��c(OH��) |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ˮ�м�������CH3COONa���壬�ٽ���ˮ�ĵ��� |
| B��25��C ʱ��pH=3�Ĵ�����pH=3��HCl��ˮ�ĵ�������������ͬ |
| C��25��Cʱ��pH=3��NH4Cl��Һ�У�ˮ�������c��H+����10-3mol/L |
| D�������¶ȣ���ˮ�������c��H+������pH���٣������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| A��ת�ƴ���Һ������ƿʱ��δϴ���ձ� |
| B����ʽ�ζ���������ˮϴ�Ӻ�ֱ��װ���� |
| C���ζ�ʱ��Ӧ��ҡ��̫���ң�������Һ�彦�� |
| D���ζ����յ�ʱ���ζ��ܼ����������� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com