
 �÷�Ӧ��ԭ��������Ϊ100%����Ӧ�Ļ�ѧ����ʽΪ________________________��
�÷�Ӧ��ԭ��������Ϊ100%����Ӧ�Ļ�ѧ����ʽΪ________________________��  �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ˮԡ |
| ˮԡ |
| ���� |
| ���� |
| ���� |
| ���� |
 �����÷�Ӧ��ԭ��������Ϊ100%����Ӧ�Ļ�ѧ����ʽΪ
�����÷�Ӧ��ԭ��������Ϊ100%����Ӧ�Ļ�ѧ����ʽΪ| ���� |
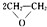
| ���� |
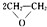
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(10��)������������ԭ�ͼ۸������ǣ���ʹ���������Ӵ�������Դ���������оƾ������ѽ���ʵ�û��Ρ�
(1)�Ҵ���ͭ�����������������£����Ա������е�����������X��X�ɷ���������Ӧ����д��X��������Һ�����ķ�Ӧ����ʽ(������巴Ӧ����)��
��
(2)�Ҵ������������ظ������Һ��Ӧ����ֱ��������Y��Y���й����ŵ�������________����Ũ��������¼��ȣ��Ҵ���Y��Ӧ������һ������ζ������W����ѧ����ʽΪ ��
(3)������ƿ��ɫҺ�壬�ֱ�ʢ��Y��W��ֻ��һ���Լ��Ϳ��Լ��𣬸��Լ�������
![]()
(4)�ִ�ʯ�ͻ�������������������ϩ�ܱ������������ɻ�������( )���÷�Ӧ��ԭ��������Ϊ100%����Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��14�֣�������������ԭ�ͼ۸������ǣ���ʹ���������Ӵ�������Դ���������оƾ������ѽ���ʵ�û��Ρ�
��1���Ҵ���ͭ�����������������£����Ա������е�����������X��X�ɷ���������Ӧ����д��X��������Һ�����ķ�Ӧ����ʽ��������巴Ӧ�������� ��
��2���Ҵ������������ظ������Һ��Ӧ����ֱ��������Y��Y ���й����ŵ�������______����Ũ���������£��Ҵ���Y��Ӧ������һ������ζ������W����ѧ����ʽΪ___________��
��3��������ƿ��ɫҺ�壬�ֱ�ʢ��Y��W��ֻ��һ���Լ��Ϳ��Լ��𣬸��Լ������� �� ��
��4���ִ�ʯ�ͻ�������������������ϩ�ܱ������������ɻ�������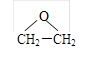 ����
����
��Ӧ��ԭ��������Ϊ100%����Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012������ʡ�����и߶���ѧ����ĩ���Ի�ѧ�Ծ��������棩 ���ͣ������
(10��)������������ԭ�ͼ۸������ǣ���ʹ���������Ӵ�������Դ���������оƾ������ѽ���ʵ�û��Ρ�
(1)�Ҵ���ͭ�����������������£����Ա������е�����������X��X�ɷ���������Ӧ����д��X��������Һ�����ķ�Ӧ����ʽ(������巴Ӧ����)��
��
(2)�Ҵ������������ظ������Һ��Ӧ����ֱ��������Y��Y���й����ŵ�������________����Ũ��������¼��ȣ��Ҵ���Y��Ӧ������һ������ζ������W����ѧ����ʽΪ ��
(3)������ƿ��ɫҺ�壬�ֱ�ʢ��Y��W��ֻ��һ���Լ��Ϳ��Լ��𣬸��Լ�������
(4)�ִ�ʯ�ͻ�������������������ϩ�ܱ������������ɻ�������( )���÷�Ӧ��ԭ��������Ϊ100%����Ӧ�Ļ�ѧ����ʽΪ
��
)���÷�Ӧ��ԭ��������Ϊ100%����Ӧ�Ļ�ѧ����ʽΪ
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�콭��ʡ�߶���ѧ�ڵ������¿���ѧ�Ծ��������棩 ���ͣ������
(10��)������������ԭ�ͼ۸������ǣ���ʹ���������Ӵ�������Դ���������оƾ������ѽ���ʵ�û��Ρ�
(1)�Ҵ���ͭ�����������������£����Ա������е�����������X��X�ɷ���������Ӧ����д��X��������Һ�����ķ�Ӧ����ʽ(������巴Ӧ����)��
��
(2)�Ҵ������������ظ������Һ��Ӧ����ֱ��������Y��Y���й����ŵ�������________����Ũ��������¼��ȣ��Ҵ���Y��Ӧ������һ������ζ������W����ѧ����ʽΪ ��
(3)������ƿ��ɫҺ�壬�ֱ�ʢ��Y��W��ֻ��һ���Լ��Ϳ��Լ��𣬸��Լ�������

(4)�ִ�ʯ�ͻ�������������������ϩ�ܱ������������ɻ�������( )���÷�Ӧ��ԭ��������Ϊ100%����Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com