
”¾ĢāÄæ”æA”«GŹĒ֊ѧ»Æѧ³£¼ūĪļÖŹ£¬A”¢DĪŖµ„ÖŹ£¬GŹĒŗ¬AŌŖĖŲµÄĖįŠŌĘųĢ唣ŅŃÖŖ£ŗA(s)+B(g)=C(g)+D(g) ¦¤H= +131.4 kJmol-1£¬Ä³Ķ¬Ń§ŹµŃé²āÖŖ£¬4 g A·¢ÉśÉĻŹö·“Ó¦ĪüŹÕ43.8 kJµÄČČĮ攣
£Ø1£©Š“³öAŌŖĖŲµÄĆū³Ę________”£
£Ø2£©ĮķĶāÖŖ£ŗ
A(s)+O2(g)=G(g)¦¤H= £393.6 kJmol-1
C(g)+ O2(g)=G(g)¦¤H=£283 kJmol-1
D(g)+ O2(g)=B(g)¦¤H=£242 kJmol-1ÓÉ“ĖÅŠ¶Ļ”°ŅņĪŖ283 kJmol-1+242 kJmol-1>393.6 kJmol-1£¬ĖłŅŌAČ¼ÉÕŹ±¼ÓÉŁĮæBæÉŅŌ·Å³öøü¶ąµÄČČĮæ”±ÕāÖÖĖµ·ØŹĒ·ńÕżČ·£æ___________£¬ĄķÓÉŹĒ_____________________£»
£Ø3£©Š“³öA+O2”śCµÄČČ»Æѧ·½³ĢŹ½£ŗ__________________________”£
”¾“š°ø”æ Ģ¼ ²»ÕżČ· 1 mol AÓėO2Ö±½ÓČ¼ÉշųöµÄČČĮæĪŖ393.6 kJ£¬¶ų1 mol AĻČÓėB·“Ӧɜ³ÉC ŗĶD£¬CŗĶDŌŁÓėO2·“Ó¦£¬¹²·ÅČČĮæ-131.4 kJ+283 kJ+242 kJ=393.6 kJ£¬Į½ÕßĻąĶ¬ C(s)+1/2O2(g)====CO(g) ¦¤H=£110.6 kJmol-1
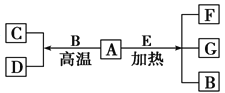
”¾½āĪö”æA”¢DĪŖµ„ÖŹ£¬GŹĒŗ¬AŌŖĖŲµÄĖįŠŌĘųĢ壬ŌņŅŖĶĘÖŖAĪŖ·Ē½šŹōŌŖĖŲ£¬A”¢DĪŖµ„ÖŹ£¬æÉÖŖAÓėBĮĖÉśĮĖÖĆ»»·“Ó¦£¬ĒŅA”¢C¶¼ÄÜÓėŃõĘų·“Ӧɜ³Éŗ¬AµÄĖįŠŌĘųĢ壬·ūŗĻ“ĖĢõ¼žµÄ֊ѧ³£¼ūµÄŌŖĖŲÓŠĢ¼ŗĶĮņ£¬µ«ÓÉÓŚAÓėBÖĆ»»µÄ²śĪļDŅ²æÉŅŌŗĶŃõĘų·“Ó¦£¬ĖłŅŌAÖ»ÄÜĪŖĢ¼ŌŖĖŲ£¬BĪŖĖ®£¬CĪŖŅ»Ńõ»ÆĢ¼£¬DĪŖĒāĘų£¬GĪŖ¶žŃõ»ÆĢ¼”£
£Ø1£©AĪŖĢ¼ŌŖĖŲ£¬¹Ź“š°øĪŖ£ŗĢ¼£»
£Ø2£©ŅņĪŖ1molAÓėO2Ö±½ÓČ¼ÉշųöµÄČČĮæĪŖ393.6kJ£¬¶ų1molAĻČÓėB·“Ӧɜ³ÉCŗĶD£¬CŗĶDŌŁÓėO2·“Ó¦£¬¹²·Å³öČČĮæ£ŗ-131.4kJ+283kJ+242kJ=393.6kJ£¬Į½ÕßĻąĶ¬£¬ĖłŅŌĢāÄæÖŠµÄĖµ·Ø²»ÕżČ·£¬¹Ź“š°øĪŖ£ŗ²»ÕżČ·£¬ŅņĪŖ1molAÓėO2Ö±½ÓČ¼ÉշųöµÄČČĮæĪŖ393.6kJ£¬¶ų1molAĻČÓėB·“Ӧɜ³ÉCŗĶD£¬CŗĶDŌŁÓėO2·“Ó¦£¬¹²·Å³öČČĮæ£ŗ-131.4kJ+283kJ+242kJ=393.6kJ£¬Į½ÕßĻąĶ¬£»
£Ø3£©C£Øs£©+![]() O2£Øg£©ØTCO£Øg£©æÉŅŌæ“×÷ŹĒ·“Ó¦¢ŁA£Øs£©+O2£Øg£©ØTG£Øg£©”÷H=-393.6kJmol-1¼õČ„·“Ó¦¢ŚC£Øg£©+
O2£Øg£©ØTCO£Øg£©æÉŅŌæ“×÷ŹĒ·“Ó¦¢ŁA£Øs£©+O2£Øg£©ØTG£Øg£©”÷H=-393.6kJmol-1¼õČ„·“Ó¦¢ŚC£Øg£©+![]() O2£Øg£©ØTG£Øg£©”÷H=-283kJmol-1µĆµ½£¬øł¾ŻøĒĖ¹¶ØĀÉæÉÖŖ£¬”÷HØT-393.6kJmol-1-£Ø-283kJmol-1£©=-110.6kJmol-1£¬¹ŹČČ»Æѧ·½³ĢŹ½ĪŖ£ŗC£Øs£©+
O2£Øg£©ØTG£Øg£©”÷H=-283kJmol-1µĆµ½£¬øł¾ŻøĒĖ¹¶ØĀÉæÉÖŖ£¬”÷HØT-393.6kJmol-1-£Ø-283kJmol-1£©=-110.6kJmol-1£¬¹ŹČČ»Æѧ·½³ĢŹ½ĪŖ£ŗC£Øs£©+![]() O2£Øg£©ØTCO£Øg£©”÷H=-110.6kJmol-1£¬¹Ź“š°øĪŖ£ŗC£Øs£©+
O2£Øg£©ØTCO£Øg£©”÷H=-110.6kJmol-1£¬¹Ź“š°øĪŖ£ŗC£Øs£©+![]() O2£Øg£©ØTCO£Øg£©”÷H=-110.6kJmol-1”£
O2£Øg£©ØTCO£Øg£©”÷H=-110.6kJmol-1”£


 ŌĶĮæģ³µĻµĮŠ“š°ø
ŌĶĮæģ³µĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æĻĀĮŠĄė×ÓÖŠ£¬Ėł“ųµēŗÉŹżÓėøĆĄė×ÓµÄŗĖĶāµē×Ó²ćŹżĻąµČµÄŹĒ£Ø £©
A.Mg2+
B.Al3+
C.Be2+
D.H+
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æŌŚ·“Ó¦KClO3£«6HCl(ÅØ)===3Cl2”ü£«KCl£«3H2OÖŠ£¬±»»¹ŌµÄĀČŌ×ÓŗĶ±»Ńõ»ÆµÄĀČŌ×ÓµÄøöŹżÖ®±ČĪŖ
A£®1:6 B£®5:1 C£®1:5 D£®6:1
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æŌŚ25”ę”¢101 kPaĢõ¼žĻĀ£¬C(s)”¢H2(g)”¢CH3COOH(l)µÄČ¼ÉÕČČ·Ö±šĪŖ393.5kJ/mol”¢285.8kJ/mol”¢870.3kJ/mol£¬Ōņ2C(s)£«2H2(g)£«O2(g)=CH3COOH(l)µÄ·“Ó¦ČČĪŖ (””””)
A£®£488.3 kJ/mol B£®£«488.3 kJ/mol
C£®£191 kJ/mol D£®£«191 kJ/mol
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”放»Æѧ”Ŗ”ŖŃ”ŠŽ3£ŗĪļÖŹ½į¹¹ÓėŠŌÖŹ”æ
£ØŅ»£©ĪųŌŖĖŲ£Øselenium£©ŹĒµŚĖÄÖÜĘŚ”¢µŚVIA×åŌŖĖŲ£¬µ„ÖŹÓŠŗģĪų”¢ŗŚĪųŗĶ»ŅĪų£¬ĘäÖŠ»ŅĪųÓŠĮ¼ŗƵĵ¼Čȵ¼µēŠŌ£¬ĒŅÓŠĻŌÖųµÄ¹āµēŠ§Ó¦£¬æÉÓ¦ÓĆÓŚ¹āµē°ėµ¼Ģå²ÄĮĻ”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©SeŌ×ÓµÄŗĖĶāµē×ÓÅŲ¼Ź½ĪŖ[Ar]_____”£
£Ø2£©AsŗĶSeŹĒĶ¬Ņ»ÖÜĘŚµÄŌŖĖŲ£¬AsµÄµŚŅ»µēĄėÄܱČSe“ó£¬ŌŅņŹĒ______”£SeO42-ÖŠSeØDOµÄ¼ü½Ē±ČSeO3µÄ¼ü½Ē________£ØĢī”°“ó”±»ņ”°Š””±£©”£
£Ø3£©H2Se±ČH2SĖ®ČÜŅŗµÄĖįŠŌĒ棬ŌŅņŹĒ_____£¬____”£
£Ø4£©»ŅĪųµÄ¾§ĢåĪŖĮł·½¾§°ū½į¹¹£¬Ō×ÓÅÅĮŠĪŖĪŽĻŽĀŻŠżĮ“£¬·Ö²¼ŌŚĮł·½¾§øńÉĻ£¬Ķ¬Ņ»ĢõĮ“ÄŚŌ×Ó×÷ÓĆŗÜĒ棬ĻąĮŚĮ“Ö®¼äŌ×Ó×÷ÓĆ½ĻČõ£¬ĘäĀŻŠżĮ“דĶ¼”¢¾§°ū½į¹¹Ķ¼ŗĶ¾§°ūø©ŹÓĶ¼ČēĻĀ”£

ĀŻŠżĮ“ÖŠSeŌ×ÓµÄŌӻƷ½Ź½ĪŖ____________”£
£Ø¶ž£©ĄūÓĆCuSO4ŗĶNaOHÖʱøµÄCu£ØOH£©2¼ģŃéČ©»łŹ±£¬Éś³ÉŗģÉ«Cu2O£¬Ę侧°ū½į¹¹ČēĶ¼ĖłŹ¾”£

¢ŁøĆ¾§°ūŌ×Ó×ų±ź²ĪŹżAĪŖ£Ø0£¬0£¬0£©£»BĪŖ£Ø1£¬0£¬0£©£»CĪŖ£Ø1/2£¬1/2£¬1/2£©”£ŌņDŌ×ÓµÄ×ų±ź²ĪŹżĪŖ_________£¬Ėü“ś±ķĶŌ×Ó”£
¢ŚČōCu2O¾§ĢåĆܶČĪŖd g”¤cm£3£¬¾§°ū²ĪŹżĪŖa pm£¬Ōņ°¢·ü¼ÓµĀĀŽ³£ŹżÖµNA=________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ¶ĢÖÜĘŚŌŖĖŲX”¢Y”¢Z”¢WŌŚŌŖĖŲÖÜĘŚ±ķÖŠµÄĻą¶ŌĪ»ÖĆČēĶ¼ĖłŹ¾£¬ĘäÖŠWŌ×ÓµÄÖŹ×ÓŹżŹĒĘä×īĶā²ćµē×ÓŹżµÄČż±¶£¬ĻĀĮŠĖµ·Ø²»ÕżČ·µÄŹĒ£Ø £©

A. Ō×Ó°ė¾¶£ŗW£¾Z£¾Y£¾X
B. ×īøß¼ŪŃõ»ÆĪļ¶ŌÓ¦Ė®»ÆĪļµÄĖįŠŌ£ŗX£¾W£¾Z
C. ×ī¼ņµ„ĘųĢ¬Ēā»ÆĪļµÄČČĪČ¶ØŠŌ£ŗY£¾X£¾W£¾Z
D. ŌŖĖŲX”¢Z”¢WµÄ×īøß»ÆŗĻ¼Ū·Ö±šÓėĘäÖ÷×åŠņŹżĻąµČ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æĻĀĮŠĪļÖŹ°“ÕÕ“æ¾»Īļ”¢»ģŗĻĪļ”¢µē½āÖŹŗĶ·Ēµē½āÖŹĖ³ŠņÅÅĮŠµÄŹĒ£Ø £©
A. ŃĪĖį”¢ĢģČ»Ęų”¢“×Ėį”¢øɱł B. µØ·Æ”¢ĘÆ°×·Ū”¢ĀČ»Æ¼Ų”¢ĀČĘų
C. ĀČ»ÆÄĘ”¢ø£¶ūĀķĮÖ”¢ĮņĖįÄĘ”¢ŅŅ“¼ D. µ„øŹÓĶõ„”¢»ģøŹÓĶõ„”¢æĮŠŌ¼Ų”¢ŹÆ»Ņ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ£Ø1£©ČĖĄąæÉŅŌÖ÷¶Æ²ĪÓėµŖŃ»·£¬ŗĻ³É°±¹¤Ņµ¾ĶŹĒ²ĪÓėµÄŹÖ¶ĪÖ®Ņ»”£ŅŌĢģČ»ĘųĪŖŌĮĻŗĻ³É°±ŹĒŠĀµÄÉś²śµŖ·ŹµÄ·½·Ø£¬Ėü¾ßÓŠĪŪČ¾Š””¢³É±¾µĶµČÖī¶ąÓÅµć”£Ęä¹ż³Ģ“óĢåČēĶ¼1ĖłŹ¾£ŗ

¢ŁĒėŠ“³öÓĆĢģČ»ĘųÖʱøĒāĘųµÄ»Æѧ·½³ĢŹ½£ŗ_________________________________£»
¢ŚŠ“³öŗĻ³ÉÄņĖŲ[CO (NH2) 2]·“Ó¦µÄ»Æѧ·½³ĢŹ½£ŗ__________________________£»
¢ŪŠ“³öO2ÓėNH3ŌŚÓŠ“߻ƼĮµÄĢõ¼žĻĀ·“Ӧɜ³ÉNH4NO3ŗĶH2OµÄ»Æѧ·½³ĢŹ½£ŗ______£»
¢ÜĆæÉś²ś1molNH4NO3×īÉŁŠčŅŖNH3______mol£¬¶ųŅŖÉś²śÕāŠ©NH3ÓÖ×īÉŁŠčŅŖCH4____mol£»
£Ø2£©æĘѧ¼ŅŅŃ»ńµĆ¼«¾ßĄķĀŪŃŠ¾æŅāŅåµÄN4·Ö×Ó£¬Ęä½į¹¹ĪŖÕżĖÄĆęĢå£ØČēĶ¼2£©£®ŅŃÖŖ¶ĻĮŃ1molN-N¼üĪüŹÕ193kJČČĮ棬¶ĻĮŃ1molN”ŌN¼üĪüŹÕ941kJČČĮ棬Ōņ1molN4ĘųĢå×Ŗ»ÆĪŖN2Ź±ŅŖŹĶ·Å______kJÄÜĮ攣
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æĻĀĮŠĖµ·ØÖŠÕżČ·µÄŹĒ£Ø £©
A.Ąė×Ó»ÆŗĻĪļÖŠæÉŅŌŗ¬ÓŠ¹²¼Ū¼ü£¬µ«¹²¼Ū»ÆŗĻĪļÖŠŅ»¶Ø²»ŗ¬Ąė×Ó¼ü
B.ŗ¬ÓŠ¹²¼Ū¼üµÄĪļÖŹŅ»¶ØŹĒ¹²¼Ū»ÆŗĻĪļ
C.HClČÜÓŚĖ®Ö»ŠčæĖ·ž·Ö×Ó¼ä×÷ÓĆĮ¦
D.øɱłÉż»ŖŹ±£¬·Ö×ÓÄŚ¹²¼Ū¼ü»į·¢Éś¶ĻĮŃ
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com