
|
ЮяжЪ |
ШлЕуЃЏЁц |
ЗаЕуЃЏЁц |
УмЖШ/gЁЄcm-3 |
|
ввДМ |
Ѓ144 |
78 |
0.789 |
|
ввЫс |
16.6 |
117.9 |
1.05 |
|
ввЫсввѕЅ |
Ѓ83.6 |
77.5 |
0.900 |
|
ХЈH2SO4 |
|
338 |
1.84 |
ЪЕбщЪвжЦввЫсввѕЅЕФжївЊзАжУШчЯТЭМ(a)ЫљЪОЃЌжївЊВНжшЮЊЃК
Ђйдк30mLЕФДѓЪдЙмжаАДЬхЛ§БШ1ЃК4ЃК4ЕФБШР§ХфжЦХЈСђЫсЁЂввДМКЭввЫсЕФЛьКЯвКЃЛ
ЂкАД(a)ЭМСЌНгзАжУЃЌЪЙВњЩњЕФеєЦјОЕМЙмЭЈЕН15mlЪдЙмЫљЪЂБЅКЭNa2CO3ШмвКЃЈМгШы1ЕЮЗгЬЊЪдвКЃЉЩЯЗН2mmЁЋ3mmДІЃЛ
ЂлаЁЛ№МгШШЪдЙмжаЕФЛьКЯвКЃЛ
ЂмД§аЁЪдЙмжаЪеМЏЕНдМ4mLВњЮяЪБЭЃжЙМгШШЃЌГЗГіаЁЪдЙмВЂгУСІеёЕДЃЌШЛКѓОВжУД§ЦфЗжВуЃЛ
ЂнЗжРыГіДПОЛЕФввЫсввѕЅЁЃ
ЧыЛиД№ЯТСаЮЪЬтЃК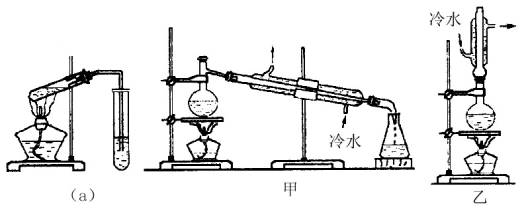
(1)ВНжшЂйжаЃЌХфжЦетвЛБШР§ЕФЛьКЯвКЕФВйзїЪЧЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁ
(2)аДГіИУЗДгІЕФЛЏбЇЗНГЬЪН________ЃЌХЈH2SO4ЕФзїгУЪЧ________ЁЃ
(3)ВНжшЂлжаЃЌгУаЁЛ№МгШШЪдЙмжаЕФЛьКЯвКЃЌЦфдвђЪЧ________ЁЃ
(4)ВНжшЂмЫљЙлВьЕНЕФЯжЯѓЪЧ________ЃЌаДГідвђгыНсТлЃК________ЁЃ
(5)ВНжшЂнжаЃЌЗжРыГіввЫсввѕЅбЁгУЕФвЧЦїЪЧ________ЃЌВњЮягІДг________ПкЕЙГіЃЌвђЮЊ________ЁЃ
(6)ЮЊЬсИпввЫсввѕЅЕФВњТЪЃЌМзЁЂввСНЮЛЭЌбЇЗжБ№ЩшМЦСЫШчЩЯЭММзЁЂввЕФзАжУЃЈввЭЌбЇД§ЗДгІЭъБЯРфШДКѓдйгУБЅКЭNa2CO3ШмвКЬсШЁЩеЦПжаВњЮяЃЉЁЃФуШЯЮЊФФжжзАжУКЯРэЃЌЮЊЪВУДЃПД№________ЁЃ
| (1)ЯШЗжБ№ЯђДѓЪдЙмжаМгШыввДМКЭввЫсИї4mLЃЛЛьКЯКѓдйЛКЛКМгШы1mLХЈH2SO4ЃЌБпМгБпеёЕДЃЌЪЙжЎЛьКЯОљдШЁЃ
(2)CH3COOH+C2H5OH (3)вђЮЊввДМЁЂввЫсЁЂввЫсввѕЅЕФЗаЕуНгНќЃЌЧвНЯЕЭЃЌДѓЛ№МгШШЃЌЗДгІЮяДѓСПеєЗЂЖјНЕЕЭВњТЪЁЃ (4)дкЧГКьЩЋNa2CO3ШмвКВуЩЯЗНгадМ4cmКёЕФЮоЩЋвКВуЁЃеёЕДКѓЃЌNa2CO3ШмвКВуЧГКьЩЋЭЪШЅЃЌгаЯИаЁЦјХнУАГіЃЌЩЯВувКЬхКёЖШБфБЁЃЛNa2CO3ШмвКгыеєГіЕФввЫсЗДгІЃЌЗХГіCO2ЃЌВЂФмЪЙввДМШмгкЫЎжаЃЈЛЙФмНЕЕЭѕЅдкЫЎжаЕФШмНтЖШЃЉДяЕНОЛЛЏВњЮяЕФФПЕФЁЃ (5)ЗжвКТЉЖЗЃЛЩЯЃЛввЫсввѕЅЕФУмЖШБШЫЎаЁЁЃ (6)ввЃЛЗДгІЮяФмОРфФ§ЛиСїЕНЩеЦПжаЃЌдіМгВњТЪЁЃ
|


 ЬьЬьЯђЩЯвЛБОКУОэЯЕСаД№АИ
ЬьЬьЯђЩЯвЛБОКУОэЯЕСаД№АИ аЁбЇЩњ10ЗжжггІгУЬтЯЕСаД№АИ
аЁбЇЩњ10ЗжжггІгУЬтЯЕСаД№АИ
| ФъМЖ | ИпжаПЮГЬ | ФъМЖ | ГѕжаПЮГЬ |
| ИпвЛ | ИпвЛУтЗбПЮГЬЭЦМіЃЁ | ГѕвЛ | ГѕвЛУтЗбПЮГЬЭЦМіЃЁ |
| ИпЖў | ИпЖўУтЗбПЮГЬЭЦМіЃЁ | ГѕЖў | ГѕЖўУтЗбПЮГЬЭЦМіЃЁ |
| ИпШ§ | ИпШ§УтЗбПЮГЬЭЦМіЃЁ | ГѕШ§ | ГѕШ§УтЗбПЮГЬЭЦМіЃЁ |
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
 ФГЛЏбЇЗДгІжаЃЌЩшЗДгІЮяЕФзмФмСПЮЊE1ЃЌЩњГЩЮяЕФзмФмСПЮЊE2ЃЎ
ФГЛЏбЇЗДгІжаЃЌЩшЗДгІЮяЕФзмФмСПЮЊE1ЃЌЩњГЩЮяЕФзмФмСПЮЊE2ЃЎ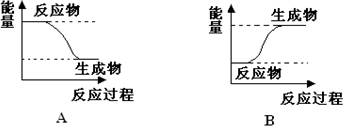
| бЮ | ШлЕу/Ёц | ШлЛЏЮќШШ/KJ?mol-1 | ВЮПММлИё/дЊ?kg-1 |
| CaCL2?6H2O | 29ЃЌ0 | 37ЃЌ3 | 780ЁЋ850 |
| Na2SO4?10H2O | 32ЃЌ4 | 77ЃЌ0 | 800ЁЋ900 |
| Na2HPO4?12H2O | 36ЃЌ1 | 100ЃЌ1 | 1600ЁЋ2000 |
| Na2S2O3?5H2O | 48ЃЌ5 | 49ЃЌ7 | 1400ЁЋ1800 |
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
| дЊЫи | Яр Йи аХ ЯЂ |
| X | XЕФФГжжЧтЛЏЮяФмЪЙЪЊШѓЕФКьЩЋЪЏШяЪджНБфРЖ |
| Y | ЕЅжЪЪЧСМКУЕФАыЕМЬхВФСЯЃЌЙуЗКгІгУгкЙтЕчаХЯЂСьгђ |
| Z | ZЕФвЛжжКЫЫижЪСПЪ§ЮЊ27ЃЌжазгЪ§ЮЊ14 |
| W | зюИпМлбѕЛЏЮяЖдгІЕФЫЎЛЏЮяЪЧвЛжжВЛШмгкЫЎЕФРЖЩЋЙЬЬх |
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
| ||
| ||
| 1 |
| 2 |
| 3 |
| 2 |
| 1 |
| 4 |
| 1 |
| 2 |
| 1 |
| 4 |
| 1 |
| 2 |
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃКдФЖСРэНт
| ЮяжЪ | ШлЕуЃЈЁцЃЉ | ЗаЕуЃЈЁцЃЉ | УмЖШЃЈg/cm3ЃЉ |
| ввДМ | -117.0 | 78.0 | 0.79 |
| ввЫс | 16.6 | 117.9 | 1.05 |
| ввЫсввѕЅ | -83.6 | 77.5 | 0.90 |
| ХЈСђЫсЃЈ98%ЃЉ | - | 338.0 | 1.84 |
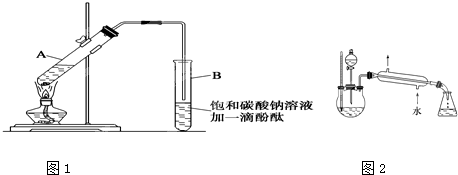
| ХЈСђЫс |
| МгШШ |
| ХЈСђЫс |
| МгШШ |
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
| AЁЂ-859 kJ?mol-1 | BЁЂ+859 kJ?mol-1 | CЁЂ-1403 kJ?mol-1 | DЁЂ-2491 kJ?mol-1 |
ВщПДД№АИКЭНтЮі>>
АйЖШжТаХ - СЗЯАВсСаБэ - ЪдЬтСаБэ
КўББЪЁЛЅСЊЭјЮЅЗЈКЭВЛСМаХЯЂОйБЈЦНЬЈ | ЭјЩЯгаКІаХЯЂОйБЈзЈЧј | ЕчаХеЉЦОйБЈзЈЧј | ЩцРњЪЗащЮожївхгаКІаХЯЂОйБЈзЈЧј | ЩцЦѓЧжШЈОйБЈзЈЧј
ЮЅЗЈКЭВЛСМаХЯЂОйБЈЕчЛАЃК027-86699610 ОйБЈгЪЯфЃК58377363@163.com