
ЎҫМвДҝЎҝПВБРКөСйІЩЧчУлФӨЖЪКөСйДҝөД»тЛщөГКөСйҪбВЫТ»ЦВөДКЗ![]()
СЎПо | КөСйІЩЧч | КөСйДҝөД»тҪбВЫ |
A | Птә¬УРЙЩБҝ | іэИҘ |
B | ПтДіИЬТәЦРјУИл | ЦӨГчИЬТәЦРә¬ |
C | ПтДіИЬТәЦРјУИлПЎСОЛбЈ¬·ЕіцОЮЙ«ОЮО¶ЖшМеЈ¬Ҫ«ЖшМеНЁИліОЗеКҜ»ТЛ®Ј¬КҜ»ТЛ®ұд»лЧЗ | ЦӨГчёГИЬТәЦРҙжФЪ |
D | Пт | ЦӨГч |
A.AB.BC.CD.
Ўҫҙр°ёЎҝA
ЎҫҪвОцЎҝ
AЈ®Птә¬УРЙЩБҝ![]() өД
өД![]() ИЬТәЦРјУИлЧгБҝ
ИЬТәЦРјУИлЧгБҝ![]() ·ЫД©Ј¬ҪБ°иТ»¶ОКұјдәу№эВЛЈ¬ҙЩҪшМъАлЧУөДЛ®ҪвЈ¬К№ЖдЧӘ»ҜОӘіБөнЈ¬ҙпөҪіэФУөДДҝөДЈ¬№КAХэИ·Ј»
·ЫД©Ј¬ҪБ°иТ»¶ОКұјдәу№эВЛЈ¬ҙЩҪшМъАлЧУөДЛ®ҪвЈ¬К№ЖдЧӘ»ҜОӘіБөнЈ¬ҙпөҪіэФУөДДҝөДЈ¬№КAХэИ·Ј»
BЈ®БтЛбұөәНAgClҫщОӘІ»ИЬУЪЛбөД°ЧЙ«іБөнЈ¬ФтПтДіИЬТәЦРјУИл![]() ИЬТәЙъіЙ°ЧЙ«іБөнЈ¬јМРшјУПЎПхЛбіБөнІ»ПыК§Ј¬ИЬТәЦРҝЙДЬә¬
ИЬТәЙъіЙ°ЧЙ«іБөнЈ¬јМРшјУПЎПхЛбіБөнІ»ПыК§Ј¬ИЬТәЦРҝЙДЬә¬![]() Ј¬№КBҙнОуЈ»
Ј¬№КBҙнОуЈ»
CЈ®ПтДіИЬТәЦРјУИлПЎСОЛбЈ¬·ЕіцОЮЙ«ОЮО¶ЖшМеЈ¬Ҫ«ЖшМеНЁИліОЗеКҜ»ТЛ®Ј¬КҜ»ТЛ®ұд»лЧЗЈ¬ЖшМеОӘ¶юСх»ҜМјЈ¬ФтОӘМјЛбСО»тМјЛбЗвСОИЬТәЈ¬№КCҙнОуЈ»
DЈ®Пт![]() ИЬТәЦРөОјУЙЩБҝЛбРФ
ИЬТәЦРөОјУЙЩБҝЛбРФ![]() ИЬТәЈ¬
ИЬТәЈ¬![]() ИЬТәНКЙ«·ўЙъСх»Ҝ»№Фӯ·ҙУҰЈ¬FeФӘЛШөД»ҜәПјЫЙэёЯЈ¬ФтЦӨГч
ИЬТәНКЙ«·ўЙъСх»Ҝ»№Фӯ·ҙУҰЈ¬FeФӘЛШөД»ҜәПјЫЙэёЯЈ¬ФтЦӨГч![]() ҫЯУР»№ФӯРФЈ¬№КDҙнОуЈ»
ҫЯУР»№ФӯРФЈ¬№КDҙнОуЈ»
ҙр°ёСЎAЎЈ


 ФД¶БҝміөПөБРҙр°ё
ФД¶БҝміөПөБРҙр°ё
| Дкј¶ | ёЯЦРҝОіМ | Дкј¶ | іхЦРҝОіМ |
| ёЯТ» | ёЯТ»Гв·СҝОіМНЖјцЈЎ | іхТ» | іхТ»Гв·СҝОіМНЖјцЈЎ |
| ёЯ¶ю | ёЯ¶юГв·СҝОіМНЖјцЈЎ | іх¶ю | іх¶юГв·СҝОіМНЖјцЈЎ |
| ёЯИэ | ёЯИэГв·СҝОіМНЖјцЈЎ | іхИэ | іхИэГв·СҝОіМНЖјцЈЎ |
ҝЖДҝЈәёЯЦР»ҜС§ АҙФҙЈә МвРНЈә
ЎҫМвДҝЎҝПВБРУР№ШЛө·ЁІ»ХэИ·өДКЗ
A.C3H8ЦРМјФӯЧУ¶јІЙУГөДКЗsp3ФУ»Ҝ
B.O2ЎўCO2ЎўN2¶јКЗ·Зј«РФ·ЦЧУ
C.ЛбРФЈәH2CO3ЈјH3PO4ЈјH2SO4ЈјHClO
D.COөДТ»ЦЦөИөзЧУМеОӘNO+Ј¬ЛьөДөзЧУКҪОӘ![]()
Ійҝҙҙр°ёәНҪвОц>>
ҝЖДҝЈәёЯЦР»ҜС§ АҙФҙЈә МвРНЈә
ЎҫМвДҝЎҝЈЁ1Ј©ПВБРФӯЧУ»тАлЧУөДөзЧУЕЕІјөДұнКҫ·Ҫ·ЁЦРЈ¬ХэИ·өДКЗ___Ј¬ОҘ·ҙБЛДЬБҝЧоөНФӯАнөДКЗ___Ј¬ОҘ·ҙәйМШ№жФтөДКЗ__ЎЈ
ўЩCa2Ј«Јә1s22s22p63s23p6
ўЪFЈӯЈә1s22s23p6
ўЫPЈә![]()
ўЬCrЈә1s22s22p63s23p63d44s2
ўЭFeЈә1s22s22p63s23p63d64s2
ўЮMg2Ј«Јә1s22s22p6
ўЯCЈә![]()
ВМЦщКҜұ»№ъјКЦйұҰҪ繫ИПОӘЛДҙуГы№уұҰКҜЦ®Т»ЎЈЦчТӘіЙ·ЦОӘBe3Al2[Si6O18]Ј¬Ттә¬ККБҝөДCr2O3ЈЁ0.15Ў«0.6%Ј©Ј¬¶шРОіЙЧжДёВМЎЈКФ»ШҙрПВБРОКМвЈә
ЈЁ2Ј©»щМ¬AlФӯЧУЦРЈ¬өзЧУМоідөДЧоёЯДЬј¶КЗ___Ј¬»щМ¬CrФӯЧУөДјЫөзЧУЕЕІјКҪКЗ___ЎЈ
ЈЁ3Ј©УГЎ°ЈҫЎұ»тЎ°ЈјЎұМоҝХЈә
өЪТ»өзАлДЬ | ·Рөг | АлЧУ°лҫ¶ |
Be___B | H2S___H2O | Al3+___O2- |
ЈЁ4Ј©ЯІЯшУлFe2Ј«әПјҙҝЙРОіЙСӘәмЛШЈ¬Fe2Ј«өДөзЧУЕЕІјКҪОӘ___Ј¬МъФЪЦЬЖЪұнЦРөДО»ЦГОӘ__ЎЈ
Ійҝҙҙр°ёәНҪвОц>>
ҝЖДҝЈәёЯЦР»ҜС§ АҙФҙЈә МвРНЈә
ЎҫМвДҝЎҝ25ЎжКұЈ¬ПаН¬pHөДБҪЦЦТ»ФӘИхЛбHAУлHBИЬТә·ЦұрјУЛ®ПЎКНЈ¬ИЬТәpHЛжјУЛ®Ме»эұд»ҜөДЗъПЯИзНјЛщКҫЎЈПВБРЛө·ЁХэИ·өДКЗ

A. HBөДЛбРФЗҝУЪHA
B. aөгИЬТәөДөјөзРФҙуУЪbөгИЬТә
C. Н¬ЕЁ¶ИөДNaAУлNaBИЬТәЦРЈ¬c(A-)=c(B-)
D. јУЛ®ПЎКНөҪpHПаН¬КұЈ¬ЛбHAЎўHBУГөҪөДЛ®V(A)РЎУЪV(B)
Ійҝҙҙр°ёәНҪвОц>>
ҝЖДҝЈәёЯЦР»ҜС§ АҙФҙЈә МвРНЈә
ЎҫМвДҝЎҝ»ҜС§УлЙъІъЎўЙъ»оГЬЗРПа№ШЎЈПВБРЛө·ЁҙнОуөДКЗ![]()
![]()
A.ЕЭДӯГр»рЖчҝЙУГУЪТ»°гөДЖр»рЈ¬ТІККУГУЪөзЖчЖр»р
B.ВМЙ«»ҜС§ТӘЗуҙУФҙН·ЙППыіэ»тјхЙЩЙъІъ»о¶Ҝ¶Ф»·ҫіөДОЫИҫ
C.№шВҜЛ®№ёЦРә¬УРөД![]() Ј¬ҝЙПИУГ
Ј¬ҝЙПИУГ![]() ИЬТәҙҰАнЈ¬әуУГЛбіэИҘ
ИЬТәҙҰАнЈ¬әуУГЛбіэИҘ
D.өзИИЛ®ЖчУГГҫ°ф·АЦ№ДЪөЁёҜКҙЈ¬ФӯАнКЗОюЙьСфј«өДТхј«ұЈ»Ө·Ё
Ійҝҙҙр°ёәНҪвОц>>
ҝЖДҝЈәёЯЦР»ҜС§ АҙФҙЈә МвРНЈә
ЎҫМвДҝЎҝМъј°Жд»ҜәПОпФЪTЕ©ТөЙъІъЦРУРЦШТӘөДЧчУГ
(1)![]() ФЪҝХЖшешЧЖЙХҝЙЦЖөГМъПөСх»ҜОпІДБПЎЈТСЦӘ
ФЪҝХЖшешЧЖЙХҝЙЦЖөГМъПөСх»ҜОпІДБПЎЈТСЦӘ![]() Ј¬101kPaКұЈә
Ј¬101kPaКұЈә
![]()
![]()
![]()
![]()
Рҙіц![]() ФЪҝХЖшЦРЧЖЙХЙъіЙ
ФЪҝХЖшЦРЧЖЙХЙъіЙ![]() өДИИ»ҜС§·ҪіМКҪ______
өДИИ»ҜС§·ҪіМКҪ______
(2)ЙъІъЦРіЈУГCO»№ФӯМъөДСх»ҜОпТұБ¶МъЎЈTТөЙПҝЙАыУГјЧНйУлЛ®ХфЖш·ҙУҰЦЖұёCOФӯБПЖшЈә![]() ЎЈИзНјКЗ
ЎЈИзНјКЗ![]() Ул
Ул![]() іхКјМе»эұИОӘ1Јә3КұәхәвМеПөЦРјЧНйөДМе»э·ЦКэКЬОВ¶ИЎўС№ЗҝУ°ПмөД№ШПөНјЈә
іхКјМе»эұИОӘ1Јә3КұәхәвМеПөЦРјЧНйөДМе»э·ЦКэКЬОВ¶ИЎўС№ЗҝУ°ПмөД№ШПөНјЈә

ўЩјЧНйУлЛ®ХфЖш·ҙУҰЦЖұёCOөД·ҙУҰОӘ______![]() МоЎ°·ЕИИЎұ»тЎ°ОьИИЎұ
МоЎ°·ЕИИЎұ»тЎ°ОьИИЎұ![]() ·ҙУҰЈ¬С№Зҝ
·ҙУҰЈ¬С№Зҝ![]() ______
______![]() МоЎ°
МоЎ°![]() Ўұ»тЎ°
Ўұ»тЎ°![]() Ўұ
Ўұ![]()
ўЪФЪәгОВәгС№өДМхјюПВЈ¬ЙПКц·ҙУҰҙпөҪЖҪәвЧҙМ¬ЎЈөұёДұд·ҙУҰөДДіТ»ёцМхјюәуЈ¬ПВБРұд»ҜДЬЛөГчЖҪәвТ»¶ЁПтДж·ҙУҰ·ҪПтТЖ¶ҜөДКЗ______
A.Дж·ҙУҰЛЩВКПИФцҙуәујхРЎ
B.»мәПЖшМеөДГЬ¶ИјхРЎ
C.»ҜС§ЖҪәвіЈКэKЦөјхРЎ
D.Л®ХфЖшөДЧӘ»ҜВКјхРЎ
ўЫФЪәгОВЎўәгИЭөДМхјюПВЈ¬ПтЙПКцЖҪәвМеПөЦРідИЛМе»эұИОӘЈә3өДјЧНйУлЛ®ХфЖш»мәПЖшМеЈ¬ФЩҙОҙпөҪЖҪәвКұCOөДМе»э·ЦКэ______![]() МоЎ°ФцҙуЎұЎўјхРЎЎұ»тЎ°І»ұдЎұ
МоЎ°ФцҙуЎұЎўјхРЎЎұ»тЎ°І»ұдЎұ![]() Ј¬
Ј¬
(3)ТСЦӘ![]() КұЈ¬
КұЈ¬![]() Ј¬ёГОВ¶ИПВ·ҙУҰ
Ј¬ёГОВ¶ИПВ·ҙУҰ![]() өДЖҪәвіЈКэ
өДЖҪәвіЈКэ![]() ______
______
(4)№ӨТөЙПНЁ№эөзҪвЕЁNaOHИЬТәҝЙЦЖұё![]() Ј¬И»әуЧӘ»ҜОӘ
Ј¬И»әуЧӘ»ҜОӘ![]() Ј¬өзҪвФӯАнИзНјЛщКҫЈ¬ФтAИЬТәЦРИЬЦКөДіЙ·ЦОӘ______
Ј¬өзҪвФӯАнИзНјЛщКҫЈ¬ФтAИЬТәЦРИЬЦКөДіЙ·ЦОӘ______![]() Мо»ҜС§КҪ
Мо»ҜС§КҪ![]() ЈәСфј«өДөзј«·ҙУҰКҪОӘ______
ЈәСфј«өДөзј«·ҙУҰКҪОӘ______
 .
.
Ійҝҙҙр°ёәНҪвОц>>
ҝЖДҝЈәёЯЦР»ҜС§ АҙФҙЈә МвРНЈә
ЎҫМвДҝЎҝТСЦӘЖшМеөДДҰ¶ыЦКБҝФҪРЎЈ¬А©ЙўЛЩ¶ИФҪҝмЎЈНјЛщКҫОӘЖшМеА©ЙўЛЩ¶ИөДКөСйЎЈБҪЦЦЖшМеА©ЙўКұРОіЙНјКҫөД°ЧЙ«СМ»·ЎЈ¶ФјЧЎўТТОпЦКөДЕР¶ПЈ¬ХэИ·өДКЗ

A.јЧКЗЕЁ°ұЛ®Ј¬ТТКЗЕЁБтЛб
B.јЧКЗЕЁ°ұЛ®Ј¬ТТКЗЕЁСОЛб
C.јЧКЗЗвСх»ҜДЖИЬТәЈ¬ТТКЗЕЁСОЛб
D.јЧКЗЕЁПхЛбЈ¬ТТКЗЕЁ°ұЛ®
Ійҝҙҙр°ёәНҪвОц>>
ҝЖДҝЈәёЯЦР»ҜС§ АҙФҙЈә МвРНЈә
ЎҫМвДҝЎҝОӘБЛјмСйФЪЗвЖшәН¶юСх»ҜМјөД»мәПЖшМеЦРКЗ·с»мИлБЛТ»Сх»ҜМјЈ¬УГИзПВөДЧ°ЦГҪшРРКөСйЎЈЗл»ШҙрЈә

(1)РҙіцұкУР·¬әЕөДТЗЖчГыіЖЈәўЩ___________Ј¬ўЪ_____________Ј¬ўЫ_____________ЎЈ
(2)Ч°ЦГBЦРУГөДКФјБКЗ _______________Ј¬ДҝөДКЗОӘБЛ_______________________ЎЈ
(3)өұ№ЫІмөҪEЧ°ЦГЦРіцПЦ____________ПЦПуКұЈ¬ЛөГч»мәПЖшМеЦРТ»¶Ёә¬УРТ»Сх»ҜМјЎЈ
(4)Из№ы»мәПЖшМеЦРә¬УРТ»Сх»ҜМјЈ¬ОӘБЛұЈ»Ө»·ҫіЈ¬УҰФЪEЧ°ЦГУТұЯөДЕЕЖш№ЬҝЪІЙИЎөДҙлК©КЗ_____ЎЈ
(5)AЧ°ЦГөДЧчУГКЗ___________Ј¬·ҙУҰөД»ҜС§·ҪіМКҪКЗ___________________ЎЈ
(6)јЩЙи»мәПЖшМеЦРөДCOУлCuOНкИ«·ҙУҰЈ¬өұНЁИлөДЖшМеОӘmgЈ¬ DФцЦШngЈ¬EЖҝФцЦШpgЎЈФт»мәПЖшМеЦРCOөДЦКБҝ°Щ·ЦКэОӘЈә_________%Ј»Из№ыИҘөфDЧ°ЦГЈ¬ХвАпјЖЛгөДCOөДЦКБҝ°Щ·ЦКэЧјИ·ВрЈҝОӘКІГҙЈҝ___________________________________________ЎЈ
Ійҝҙҙр°ёәНҪвОц>>
ҝЖДҝЈәёЯЦР»ҜС§ АҙФҙЈә МвРНЈә
ЎҫМвДҝЎҝөзөјВККЗәвБҝөзҪвЦКИЬТәөјөзДЬБҰҙуРЎөДОпАнБҝЈ¬ҫЭИЬТәөзөјВКұд»ҜҝЙТФИ·¶ЁөО¶Ё·ҙУҰөДЦХөгЎЈУТНјКЗДіН¬С§УГ0.1molL-1KOHИЬТә·ЦұрөО¶ЁМе»эҫщОӘ20mLЎўЕЁ¶ИҫщОӘ0.1molL-1өДHClәНCH3COOHИЬТәөО¶ЁЗъПЯКҫТвНј(»мәПИЬТәМе»эұд»ҜәцВФІ»јЖ)Ј®ПВБРУР№ШЕР¶ПІ»ХэИ·өДКЗ
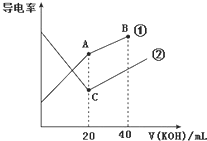
AЈ®ЗъПЯўЩҙъұн0.1 molL-1 KOHИЬТәөО¶ЁCH3COOHИЬТәөДөО¶ЁЗъПЯ
BЈ®ФЪAөгөДИЬТәЦРУРЈәc(CH3COO-)+c(OH-)-c(H+)ЁT0.05 molL-1
CЈ®ФЪBөгөДИЬТәЦРУРЈәc(K+)Јҫc(OH-)Јҫc(CH3COO-)Јҫc(H+)
DЈ®ФЪПаН¬ОВ¶ИПВЈ¬CөгЛ®өзАлөДc(H+)ҙуУЪAөгЛ®өзАлөДc(H+)
Ійҝҙҙр°ёәНҪвОц>>
°Щ¶ИЦВРЕ - Б·П°ІбБРұн - КФМвБРұн
әюұұКЎ»ҘБӘНшОҘ·ЁәНІ»БјРЕПўҫЩұЁЖҪМЁ | НшЙПУРәҰРЕПўҫЩұЁЧЁЗш | өзРЕХ©ЖӯҫЩұЁЧЁЗш | ЙжАъК·РйОЮЦчТеУРәҰРЕПўҫЩұЁЧЁЗш | ЙжЖуЗЦИЁҫЩұЁЧЁЗш
ОҘ·ЁәНІ»БјРЕПўҫЩұЁөз»°Јә027-86699610 ҫЩұЁУКПдЈә58377363@163.com