
| | A | B | C | D | E | F | G | H | I | J |
| Ō×Ó°ė¾¶(10£10m) | 0.74 | 1.60 | 1.52 | 1.10 | 0.99 | 1.86 | 0.75 | 0.82 | 0.102 | 0.037 |
| ×īøß»ņ×īµĶ »ÆŗĻ¼Ū | | £«2 | £«1 | £«5 | £«7 | £«1 | £«5 | £«3 | +6 | +1 |
| £2 | | | £3 | £1 | | £3 | | -2 | |

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
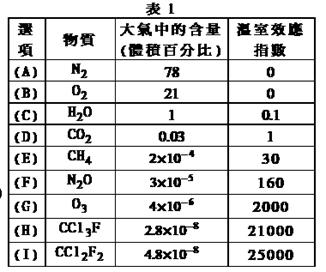
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®NaOH”¢KOH”¢CsOH¼īŠŌŅĄ“ĪŌöĒæ |
| B£®Li”¢Na”¢K”¢Rb”¢Csµ„ÖŹµÄŹ§µē×ÓÄÜĮ¦Öš½„ŌöĒæ |
| C£®Al”¢Mg”¢NaµÄŌ×Ó°ė¾¶ŅĄ“Ī¼õŠ” |
| D£®HCl”¢H2S”¢PH3ĪČ¶ØŠŌŅĄ“Ī¼õČõ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®ĖįŠŌH2CO3>H2SiO3 | B£®¼īŠŌNaOH>Mg(OH)2 |
| C£®ČČĪČ¶ØŠŌ H2O>H2S | D£®ĆÜ¶Č£ŗ±½>Ė® |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®¢÷A×åŌŖĖŲŹĒĶ¬ÖÜĘŚÖŠ·Ē½šŹōŠŌ×īĒæµÄŌŖĖŲ |
| B£®µŚ¶žÖÜĘŚ¢ōA×åŌŖĖŲµÄŌ×ÓŗĖµēŗÉŹżŗĶÖŠ×ÓŹż·Ö±šĪŖ6 |
| C£®Ö÷×åŌŖĖŲµÄ×īøßÕż¼Ū¾łµČÓŚĘä×åŠņŹż |
| D£®·Ē½šŹōŌŖĖŲµÄĘųĢ¬Ēā»ÆĪļČÜÓŚĖ®ŗó£¬Ė®ČÜŅŗ¾łĪŖĖįŠŌ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
 | ¢ńA | ¢ņA | ¢óA | ¢ōA | ¢õA | ¢öA | ¢÷A | 0 |
| 2 | | | | | | ¢Ž | ¢ß | |
| 3 | ¢Ł | ¢Ū | ¢Ż | | | | | ¢ą |
| 4 | ¢Ś | ¢Ü | | | | | | |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®µŚ¢÷A×åµÄĀ±ĖŲ | B£®Ö÷×åŌŖĖŲ |
| C£®ø±×åŌŖĖŲ | D£®µ„ÖŹÓŠŗÜĒæµÄŃõ»ÆŠŌ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®ÄĘŌ×ÓŹ§Č„Ņ»øöµē×Óŗó£¬ĖüµÄµē×ÓŹżÓėÄŹŌ×ÓĻąĶ¬£¬ĖłŅŌ±ä³ÉÄŹŌ×Ó |
| B£®ČĪŗĪŌŖĖŲµÄŌ×Ó¶¼ŹĒÓÉŗĖĶāµē×ÓŗĶŗĖÄŚÖŠ×Ó”¢ÖŹ×Ó×é³ÉµÄ |
| C£®ŗĖĖŲÖøŅ»ÖÖŌ×Ó£¬¶ųŌŖĖŲŹĒÖøŅ»ĄąŌ×Ó£»ŗĖĖŲ¼ä»„³ĘĶ¬Ī»ĖŲ£¬ŌŖĖŲ°üŗ¬Ķ¬Ī»ĖŲ |
| D£®40 18Ar”¢40 19K”¢40 20CaµÄÖŹĮæŹżĻąĶ¬£¬ĖłŅŌĖüĆĒŹĒ»„ĪŖĶ¬Ī»ĖŲ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
 ŠŌŅģ¹¹
ŠŌŅģ¹¹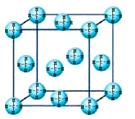
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com