
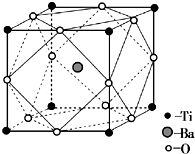
 �ѱ���Ϊ��δ�����͵Ľ��������Իش��������⣺
�ѱ���Ϊ��δ�����͵Ľ��������Իش��������⣺| 1 |
| 8 |
| 1 |
| 2 |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A�����ζԳ� |
| B���Զ��Գ� |
| C������ֵ��x��y��z���ϵķĴ��� |
| D�����ഹֱ�Ļ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
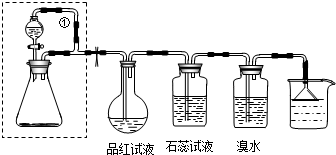
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��ij������Һ��ֻ��NH4+��Cl-��H+��OH-�������ӣ���Һ��һ�����ڣ�c��Cl-����c��NH4+����c��H+����c��OH-�� |
| B�����ʵ���Ũ����ͬ��4����Һ����CH3COONa����NaNO3����Na2CO3����NaOH��pH�Ĵ�С˳���ǣ��ܣ��ۣ��٣��� |
| C����Na2CO3��NaHCO3�Ļ����Һ�У�c��Na+��+c��H+��=c��HCO3-��+c��OH-��+c��CO32-�� |
| D��25��ʱ��pH=10��CH3COONa��Һ��pH=10�İ�ˮ�У���ˮ�������c��OH-��֮��Ϊ1��1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
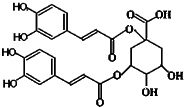 ������һ���½ṹ���͵Ŀ������ײ����Ϳ����̲������Ļ������ṹ��ͼ��ʾ���й����ص�˵����ȷ���ǣ����ĸ���ͬ���ŵ�̼��������̼����������
������һ���½ṹ���͵Ŀ������ײ����Ϳ����̲������Ļ������ṹ��ͼ��ʾ���й����ص�˵����ȷ���ǣ����ĸ���ͬ���ŵ�̼��������̼����������| A�������к���6������̼ԭ�� |
| B��һ���������ܷ���������Ӧ����ȥ��Ӧ |
| C���������Ȼ�����Һ������ɫ��Ӧ |
| D��1 mol����������11 mol NaOH��Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��0.1mol/L NaHCO3��Һ�У�c��H+��+c��Na+��=c��OH-��+c��HCO3-��+c��CO32-�� |
B��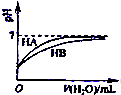 ��ʾ�����£�ϡ��HA��HB�������ϡ��Һʱ����ҺPH���ˮ���ı仯����NaA��Һ��PH����ͬŨ�ȵ�NaB��Һ��PH |
C�� ��ʾ������������ͬʱ���ֱ���T1��T2�¶�����CO2��H2�ϳɼ״������ʵ�����ʱ��仯�������CO2��H2�ϳɼ״��Ƿ��ȷ�Ӧ |
D�� ��ʾ��1.000 mol/L��ˮ����20.00mL1.000 mol/L�����У���ҺPH���¶�����백ˮ����仯���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ���� | ���� | ���� |
| �� | ��pH��ֽ���� | ��Һ��pH����7 |
| �� | ����Һ�еμ���ˮ���ټ���CCl4������ | CCl4����Ϻ�ɫ |
| �� | ������ˮ��Һ�м���Ba��NO3��2��Һ��ϡHNO3 | �а�ɫ�������� |
| �� | ���ˣ�����Һ�м���AgNO3��Һ��ϡHNO3 | �а�ɫ�������� |
| A���϶����е������Ǣۢܢ� |
| B���϶�û�е������Ǣڢ� |
| C������ȷ���������Ǣۢ� |
| D�����ܺ��е������Ǣ٢� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 һ���л���Ļ�ѧʽΪC4H4�����ӽṹ��ͼ��ʾ�������л���������������Ϻ���գ����ɵ�±����������У�������
һ���л���Ļ�ѧʽΪC4H4�����ӽṹ��ͼ��ʾ�������л���������������Ϻ���գ����ɵ�±����������У�������| A��2 | B��4 | C��5 | D��6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��NaI��NaIO3��һ���������ܷ�Ӧ����I2 |
| B��I2�ڷ�Ӧ�����ǻ�ԭ����ڷ�Ӧ�������������� |
| C��������Ӧ�����ɵ�����I2ʱת�Ƶ�������� |
| D�������ԣ�Mn02��SO42-��I03-��I2 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com