
| “߻ƼĮ | ”÷ |

| 1000¦Ńw |
| M |
| 1000”Į0.91”Į25% |
| 17 |
| amol |
| 4 |
| Ņ»¶ØĢõ¼ž |
| 4L”Į5 |
| 4”Į0.20 |
| 4L |
| 4L+25L |
| 5 |
| 4 |
| 1.5mol |
| 20.0mol |
| 18.0+1.5 |
| 20 |
| “߻ƼĮ |
| ”÷ |
| ¢Ł+¢Ś |
| 2 |
| 5 |
| 4 |
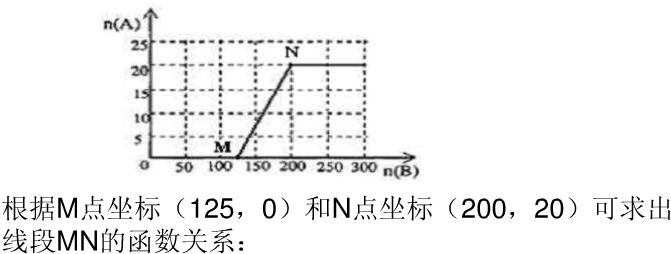 £¬
£¬ £®
£®

 ŌĶĮæģ³µĻµĮŠ“š°ø
ŌĶĮæģ³µĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012-2013ѧğÉĻŗ£ŹŠĒąĘÖĒųøßČżÉĻѧʌʌĩ½Ģѧ֏Įæµ÷ŃŠ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗ¼ĘĖćĢā
£Ø1£©°±ĘųŌŚ¹¤Å©ŅµÓŠ×Źć·ŗµÄÓĆĶ¾”£ŅŃÖŖ25%°±Ė®µÄĆܶČĪŖ0.91 g/cm3£¬5%°±Ė®µÄĆܶČĪŖ0.98 g/cm3”£
¢ŁÅäÖĘ100mL 2.5mol/L°±Ė®ŠčŅŖÅضČĪŖ25%°±Ė®______mL£Ø±£Įō2Ī»Š”Źż£©”£
¢ŚČō½«ÉĻŹöĮ½ČÜŅŗµČĢå»ż»ģŗĻ£¬ĖłµĆ°±Ė®ČÜŅŗµÄÖŹĮæ·ÖŹżŹĒ_____________”£
A£®µČÓŚ15% B£®“óÓŚ15% C£®Š”ÓŚ15% D£®ĪŽ·Ø¹ĄĖć
ŅŃÖŖ£ŗ4NH3+O2 4NO+6 H2O£¬4NO+3O2+2H2O
4NO+6 H2O£¬4NO+3O2+2H2O 4HNO3
4HNO3
£Ø2£©ÉčæÕĘųÖŠŃõĘųµÄĢå»ż·ÖŹżĪŖ0.20£¬µŖĘųµÄĢå»ż·ÖŹżĪŖ0.80”£
¢Ła mol NOĶźČ«×Ŗ»ÆĪŖHNO3ĄķĀŪÉĻŠčŅŖŃõĘų_____________mol”£
¢ŚĪŖŹ¹NH3Ē”ŗĆĶźČ«Ńõ»ÆĪŖNO£¬°±£æÕĘų»ģŗĻĘųĢåÖŠ°±µÄĢå»ż·ÖŹż£ØÓĆŠ”Źż±ķŹ¾£©ĪŖ_____________
£Ø±£Įō2Ī»Š”Źż£©”£
£Ø3£©20.0 mol NH3ÓĆæÕĘųŃõ»Æ£¬²śÉś»ģŗĻĪļµÄ×é³ÉĪŖ£ŗNO 18.0 mol”¢O2 12.0 mol”¢N2 150.0 molŗĶŅ»¶ØĮæĻõĖį£¬ŅŌ¼°ĘäĖū³É·Ö£ØøßĪĀĻĀNOÓėO2²»»ÆŗĻ£©”£¼ĘĖć°±×Ŗ»ÆĪŖNOŗĶHNO3µÄ×Ŗ»ÆĀŹ”£
£Ø4£©20.0 mol NH3ŗĶŅ»¶ØĮææÕĘų³ä·Ö·“Ó¦ŗó£¬ŌŁ×Ŗ»ÆĪŖĻõĖį”£Ķعż¼ĘĖć£¬ŌŚĶ¼ÖŠ»³öHNO3µÄĪļÖŹµÄĮæn(A)ŗĶæÕĘųµÄĪļÖŹµÄĮæn (B)¹ŲĻµµÄĄķĀŪĒśĻß”£

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012-2013ѧğÉĻŗ£ŹŠĒąĘÖĒųøßæ¼Ņ»Ä£»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
°±ĘųŌŚ¹¤Å©ŅµÓŠ×Źć·ŗµÄÓĆĶ¾”£ŅŃÖŖ25%°±Ė®µÄĆܶČĪŖ0.91 g/cm3£¬5%°±Ė®µÄĆܶČĪŖ0.98 g/cm3”£
1”¢£Ø1£©ÅäÖĘ100mL 2.5mol/L°±Ė®ŠčŅŖÅضČĪŖ25%°±Ė®______mL£Ø±£Įō2Ī»Š”Źż£©”£
£Ø2£©Čō½«ÉĻŹöĮ½ČÜŅŗµČĢå»ż»ģŗĻ£¬ĖłµĆ°±Ė®ČÜŅŗµÄÖŹĮæ·ÖŹżŹĒ_____________”£
A£®µČÓŚ15% B£®“óÓŚ15% C£®Š”ÓŚ15% D£®ĪŽ·Ø¹ĄĖć
2”¢ŅŃÖŖ£ŗ4NH3+O2 4NO+6NO£¬4NO+3O2+2H2O
4NO+6NO£¬4NO+3O2+2H2O 4HNO3
4HNO3
ÉčæÕĘųÖŠŃõĘųµÄĢå»ż·ÖŹżĪŖ0.20£¬µŖĘųµÄĢå»ż·ÖŹżĪŖ0.80”£
£Ø1£©a mol NOĶźČ«×Ŗ»ÆĪŖHNO3ĄķĀŪÉĻŠčŅŖŃõĘų_____________mol”£
£Ø2£©ĪŖŹ¹NH3Ē”ŗĆĶźČ«Ńõ»ÆĪŖNO£¬°±-æÕĘų»ģŗĻĘųĢåÖŠ°±µÄĢå»ż·ÖŹż£ØÓĆŠ”Źż±ķŹ¾£©ĪŖ_____________ £Ø±£Įō2Ī»Š”Źż£©”£
£Ø3£©20.0 mol NH3ÓĆæÕĘųŃõ»Æ£¬²śÉś»ģŗĻĪļµÄ×é³ÉĪŖ£ŗNO 18.0 mol”¢O2 12.0 mol”¢N2 150.0 molŗĶŅ»¶ØĮæĻõĖį£¬ŅŌ¼°ĘäĖū³É·Ö£ØøßĪĀĻĀNOÓėO2²»»ÆŗĻ£©”£¼ĘĖć°±×Ŗ»ÆĪŖNOŗĶHNO3µÄ×Ŗ»ÆĀŹ”£
£Ø4£©20.0 mol NH3ŗĶŅ»¶ØĮææÕĘų³ä·Ö·“Ó¦ŗó£¬ŌŁ×Ŗ»ÆĪŖĻõĖį”£Ķعż¼ĘĖć£¬ŌŚĶ¼ÖŠ»³öHNO3µÄĪļÖŹµÄĮæn(A)ŗĶæÕĘųµÄĪļÖŹµÄĮæn (B)¹ŲĻµµÄĄķĀŪĒśĻß”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
 4NO+6H2O£¬4NO+3O2+2H2O”ś4HNO3
4NO+6H2O£¬4NO+3O2+2H2O”ś4HNO3
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012-2013ѧğÉĻŗ£ŹŠĒąĘÖĒųøßČż£ØÉĻ£©ĘŚÄ©ōßøßæ¼»ÆѧŅ»Ä£ŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗ½ā“šĢā
 4NO+6H2O£¬4NO+3O2+2H2O”ś4HNO3
4NO+6H2O£¬4NO+3O2+2H2O”ś4HNO3
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com