
ij�¶��£�Ag2SO4��ˮ�еij����ܽ�ƽ��������ͼ��ʾ������˵����ȷ����
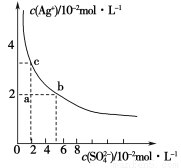
A��a���ʾAg2SO4�IJ�������Һ������Na2SO4���岻��ʹ��Һ��a��䵽b��
B��a���ʾAg2SO4�IJ�������Һ����������ʹ��Һ��a��䵽c��
C��0.04 mol��L��1��AgNO3��Һ��0.2 mol��L��1��Na2SO4��Һ����������Ag2SO4��������
D��Ag2SO4���ܶȻ�����(Ksp)Ϊ1��10��3
 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2017�츣��ʡ������ٽ��Կ��Ի�ѧ�Ծ��������棩 ���ͣ�ʵ����
(1)�á�84����Һ�������ʵ���Ũ��ԼΪ________mol��L��1��
84����Һ
��Ч�ɷ� NaClO
��� 1 000 mL
�������� 25%
�ܶ� 1.19 g��cm��3
(2)ijͬѧȡ100mL�á�84����Һ����ϡ�ͺ�����������ϡ�ͺ����Һ��c(Na��)��______mol��L��1��
(3)��ͬѧ���ĸá�84����Һ�����䷽������NaClO��������480mL��NaClO��������Ϊ25%������Һ������˵����ȷ����________(����ĸ)��
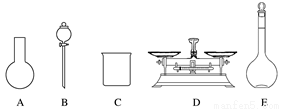
A����ͼ��ʾ�������У��������Dz���Ҫ�ģ�����Ҫһ�ֲ�������
B������ƿ������ˮϴ����Ӧ��ɺ����������Һ����
C�����ƹ����У�δ������ˮϴ���ձ��Ͳ��������ܵ��½��ƫ��
D����Ҫ����NaClO���������Ϊ143.0g
(4)��84����Һ����ϡ������ʹ�ÿ���ǿ����������ij����С����Ա��98%(�ܶ�Ϊ1.84g��cm��3)��Ũ��������2L 2.3mol��L��1��ϡ����������ǿ��84����Һ��������������
�������Ƶ�ϡ�����У�H�������ʵ���Ũ��Ϊ________mol��L��1��
������Ũ��������Ϊ________mL��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017�����ʡ�����и����ϵڶ����¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
�����йػ�ѧ���������ȷ����
A���Ҵ��ķ���ʽ��CH3CH2OH B��������ĵ���ʽ��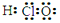
C��NH4Cl�ĵ���ʽ�� D����ȩ�ṹ��ʽ��CH3CHO
D����ȩ�ṹ��ʽ��CH3CHO
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�����ʡ��һ�ϵ�һ���ʼ컯ѧ�Ծ��������棩 ���ͣ�ѡ����
NAΪ�����ӵ�������ֵ������������ȷ����
A��NA��CO2����ռ�е����һ��Ϊ22.4L
B����״���£�22.4L���Ȼ�̼���еķ�����ΪNA
C�����³�ѹ�£�1.06g Na2CO3���е�Na+��Ϊ0.02 NA
D�����ʵ���Ũ��Ϊ0.5mol/L��MgCl2��Һ�У���Cl-����ΪNA
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�����ʡ��һ�ϵ�һ���ʼ컯ѧ�Ծ��������棩 ���ͣ�ѡ����
��ʢ��Ũ�����Լ�ƿ�ı�ǩ��Ӧӡ�����о�ʾ����е�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�꼪��ʡ�߶���9���¿���ѧ���������棩 ���ͣ�ѡ����
25��ʱ���������ˮ�зֱ�����������������ʣ�����ʵ�������±���ʾ��
�������� | HCl(aq) | FeCl3 | NaOH | CH3COONa | C2H5OH |
��Һ��pH | 3 | 4 | 10 | 11 | δ�ⶨ |
ˮ�ĵ���̶� | ��1 | ��2 | ��3 | ��4 | ��5 |
�������ʺ�ˮ�ĵ���̶��ɴ�С����˳����ȷ����
A����3>��1>��5>��4>��2 B����4>��2>��5>��3>��1
C����2>��4>��5>��1>��3 D����1>��3>��5>��2>��4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�꼪��ʡ�߶���9���¿���ѧ���������棩 ���ͣ�ѡ����
���и�����������Һ��һ���ܴ����������
A��Fe3+��NH4+��Cl����I�� B��Cu2+��Na+��Cl����NO3��
C��K+��HCO3����OH����Ba2+ D��K+��CO32����NO3����Al3+
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ��ɽ��ʡ�߶���9���¿���ѧ���������棩 ���ͣ�ѡ����
��ѧ�����������Ź㷺��Ӧ�ã����ж�Ӧ��ϵ�������
��ѧ���� | ʵ��Ӧ�� | |
A. | Al2(SO4)3��С�մ�Ӧ | ��ĭ�������� |
B. | ����ͭ������ǿ | FeCl3��ʴCu����ӡˢ��·�� |
C. | �������ξ��������� | Ư��Ư��֯�� |
D. | HF��SiO2��Ӧ | ������ڲ��������Ͽ�ʴ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017�켪��ʡ������ѧ��9���¿���ѧ�Ծ��������棩 ���ͣ������
NaNO2��ۺ�ʳ�����ƣ�������ζ������ʹ����ʳ�ж�����֪NaNO2�ܷ������·�Ӧ��2NaNO2 ��4HI = 2NO�� �� I2 �� 2NaI �� 2H2O��
��������Ӧ����������___________��
�Ƹ���������Ӧ������NaNO2��NaCl����ѡ�õ������У��ٵ⻯�ص�����ֽ �ڵ��� �۰� ��ʳ�ף�����ΪӦѡ�õ�������________������ţ���
��ij����Һ�У�����2%��5%��NaNO2��ֱ���ŷŻ������Ⱦ�������Լ���ʹNaNO2ת��Ϊ�����������Ⱦ��N2����________��
A��NaCl B��NH4Cl C��HNO3 D��ŨH2SO4
������ƽ���»�ѧ����ʽ Al�� NaNO3�� NaOH �� NaAlO2�� N2���� H2O������Ӧ������ת��5 mol���ӣ������ɱ�״����N2�����Ϊ________L��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com