
���ԭ���ڻ�ѧ��ҵ���й㷺Ӧ�á���ͼ��ʾһ�����أ�װ�е��Һa��X��Y������缫�壬ͨ��������ֱ����Դ������
��ش��������⣺
��1����X��Y���Ƕ��Ե缫��a�DZ���NaCl��Һ��ʵ�鿪ʼʱ��ͬʱ�����߸����뼸�η�̪��Һ����
�ٵ�����X���ϵĵ缫��ӦʽΪ ��
��X�������۲쵽�������� ����
��Y�缫�ϵĵ缫��ӦʽΪ ��
����õ缫��Ӧ����ķ����� ��
��2����Ҫ�õ�ⷽ��������ͭ�����Һaѡ��CuSO4��Һ����
��X�缫�IJ����� ���缫��Ӧʽ�� ��
��Y�缫�IJ����� ���缫��Ӧʽ�� ��
��˵�������ʷ����ĵ缫��Ӧ����д����
�Ţ�2H+ + 2e - = H2����2�֣� �ų����壬��Һ��졣��2�֣�
��2Cl - - 2e - = Cl2 ����2�֣�
��ʪ��ĵ⻯�ص�����ֽ����Y�缫��������ֽ����ɫ����2�֣�
�Ƣٴ�ͭ����ͭ����1�֣� Cu2+ + 2e - =" Cu" ��2�֣�
�ڴ�ͭ��1�֣� Cu - 2e - = Cu2+ ��2�֣�
���������������1��X��Ϊ�����������ӷŵ磬������������X��������������ˮ������������������ӣ�������̪����Һ��죻Y��Ϊ�����������ӷŵ磬���������������ļ��鷽��Ϊ����ʪ��ĵ⻯�ص�����ֽ����Y�缫��������ֽ����ɫ����2��������ͭ������Ϊ��ͭ����X����������ԭ��Ӧ���缫��ӦʽΪCu2+ + 2e - = Cu������Ϊ��ͭ����Y��������������Ӧ���缫��ӦʽΪCu - 2e - = Cu2+��
���㣺����
����������������߿�����֪ʶ�㣬ע�ؿ���ѧ���������⡢��������������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijʵ��С�����ݼ״�ȼ�յķ�Ӧԭ���������ͼ��ʾ��װ�á���֪�׳ص��ܷ�ӦʽΪ��2CH3OH+3O2+4KOH=2K2CO3+6H2O����ش�
��ͨ��O2�ĵ缫������ ��B�缫�������� ��
��ͨ��CH3OH�ĵ缫�ĵ缫��Ӧʽ�� ��A�缫�ĵ缫��ӦʽΪ ��
���ҳ����Ϊ1L����AgNO3����������µ��һ��ʱ�����Һ��PH��Ϊ1���������ʱ����ת�Ƶĵ�����ĿΪ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ˮ�Ƿḻ����Դ���⣬�Ӻ�ˮ����ȡԪ���ǻ�ѧ��ҵ����Ҫ��ɲ��֡�
��1�����ξ��ƾ��dz�ȥ���е�Ca2+��Fe3+��SO42-����ɳ�����ʣ��������Լ��У���Na2CO3��Һ ��HCl�����ᣩ ��Ba��OH��2��Һ���������Լ�������˳����_________������ţ���
��2�����������С���ˮ���塱����ȡ�嵥�ʷ�Ӧ�����ӷ���ʽΪ��__________��
��3��ijͬѧ�������ͼװ�ý������µ绯ѧʵ�顣
�ٵ�����K��a����ʱ�������������ݲ�����������Ϊ_______����
��һ��ʱ���ʹ����K��a�Ͽ�����b����ʱ�����߿��ڵ�װ�ÿɳ�Ϊ__________����д����ʱFe�缫�ϵĵ缫��Ӧʽ_________________��
��4��ij������ʢ��CaSO4����Һ�ķ�Ӧ����ͨ�백������ȡ���ʣ�NH4��2SO4��Ч�����á���ͨ��CO2��������������NH4��2SO4���������ԭ�� ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ͼװ���У�b�缫�ý��� M�Ƴɣ�a��c��dΪʯī�缫����ͨ��Դ������M������b����ͬʱa��d�缫�ϲ������ݡ��Իش�
��1��aΪ ����c���ĵ缫��ӦʽΪ ��
��2��������һ��ʱ�������c���ϵ��Թ���Ҳ���ռ��������壬��ʱc���ϵĵ缫��ӦʽΪ ��
��3����d�����ռ���44.8mL���壨��״����ʱֹͣ��⣬a���Ϸų�����������ʵ���Ϊ ����b�缫�ϳ�������M������Ϊ0.432g����˽�����Ħ������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪��1molH2��Ҫ����436kJ��������1molO2��Ҫ����496kJ�������γ�ˮ�����е�1molH��O�ܹ��ͷ�463kJ�����������������������ݼ��㷴Ӧ��
2H2(g)+O2(g)=2H2O(g) ����H = ��
��2����ͼ��ʾ�����γ�����ȼ�ϵ�ء�ͨ������ȼ�ϵ������ʽ(���������ҺΪH2SO4��Һʱ)�ͼ�ʽ[���������ҺΪNaOH(aq)��KOH(aq)ʱ]���֡��Իش��������⣺
����ʽ��صĵ缫��Ӧ������________________������______________������ܷ�Ӧ��______________���������ҺpH�ı仯________(��������С�����䡱)��
�ڼ�ʽ��صĵ缫��Ӧ������________________������______________������ܷ�Ӧ��______________���������ҺpH�ı仯________(��������С�����䡱)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��25��ʱ����ʯī�缫��� ��
�� ��Һ������
��Һ������ ���ӷ���ת�ƣ��Իش��������⣺
���ӷ���ת�ƣ��Իش��������⣺
��1���������� ��Ӧ���缫��ӦΪ ��
��2���������� ��Ӧ���缫��ӦΪ ��
��3������õ��� �������� ���õ�
�������� ���õ� ���������״������ ��
���������״������ ��
��4�����õ�����������ͭƬ����ʯī���缫��������ͭƬ��������� �����Һ�� �����С����������䡱��
�����С����������䡱��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
����ʹ�ò��ϣ������������
�� ���������������������зdz���Ҫ�IJ��ϣ���ÿ������ʴ����ʧ������ʮ�־��ˡ�������ʴ��Ϊ��ѧ��ʴ�͵绯ѧ��ʴ�����ߵ��۱��ʶ��Ƿ��� �Ĺ��̡�Ϊ��ֹ�ִ������ں�ˮ�б���ʴ��һ���ڴ������� ��ѡ�п�顱��ͭ�顱����
�� ����ͨ������������У�̼�������������ڸ����·�����Ӧ�Ļ�ѧ����ʽ��
������������̥����Ҫԭ�ϣ���Ȼ��ͨ����ʩ��ʹ���ķ���ת��Ϊ �ṹ���Ӷ�������ǿ�ȡ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
�����̿�MnCO3��������Fe2O3��FeO��HgCO3��2HgO�����ʣ���ҵ�������̿���ȡ�̣������������£�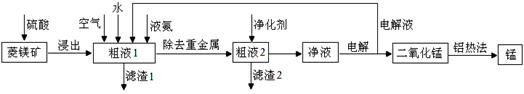
��ش��������⣺
��1�����Һ1�м����ˮ�����Ҫ �������ܴﵽ����Ҫ��
��2���������õĿ�������Ĥ���뷨�Ʊ��ĸ����������÷�����ԭ���� ��
��3����������Ҫ�ɷ�Ϊ(NH4)2S����Һ2�з�����Ҫ��Ӧ�����ӷ���ʽΪ ��
��4��д�������ĵ缫��Ӧʽ ��˵�����Һѭ����ԭ�� ��
��5��д�����ȷ����̵Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�±���ʵ������ܴﵽʵ��Ŀ�ĵ���
| | ʵ����� | ʵ��Ŀ�� |
| A | �ӵı�����Һ�еμ�ϡ��ˮ | ��֤���屽��Ϊ��ɫ���� |
| B | ���������Һ�м����Ƶ�Cu(OH)2����Һ������ | ȷ���������к���ȩ�� |
| C | ��ƾ�������Ļ��Һ�м�������� | ȷ���ƾ��л��д��� |
| D | ��������������������Һ����һ��ʱ�䣬������ȴ��Ļ��Һ�еμ���������Һ | ����ˮ������е������� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com