
| Ũ���� |
| �� |
| Ũ���� |
| �� |
| Ũ���� |
| �� |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
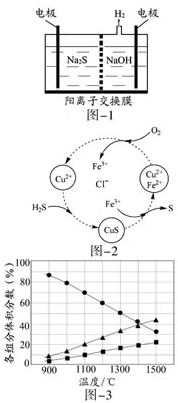 �����ת������Դ���úͻ�����������Ҫ�о����⣬�������������ж��ַ�����
�����ת������Դ���úͻ�����������Ҫ�о����⣬�������������ж��ַ������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A-B | A=B | A��B | |
| CO | 357.7 | 798.9 | 1071.9 |
| N2 | 154.8 | 418.4 | 941.7 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
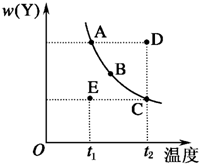 ����2X��g��?Y��g���ķ�Ӧ��ϵ����ѹǿһ��ʱ��ƽ����ϵ��Y����������w��Y�����¶ȵı仯�����ͼ��ʾ��ʵ���ϵ��κ�һ��Ϊ��Ӧ�¶��µ�ƽ��״̬����
����2X��g��?Y��g���ķ�Ӧ��ϵ����ѹǿһ��ʱ��ƽ����ϵ��Y����������w��Y�����¶ȵı仯�����ͼ��ʾ��ʵ���ϵ��κ�һ��Ϊ��Ӧ�¶��µ�ƽ��״̬�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A����ֽ | B��ʯ�� |
| C���������� | D�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
A����Һ��
| ||
| B����Һ��c��H+������ | ||
| C����Һ�ĵ���������С | ||
| D��CH3COOH�ĵ���̶����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
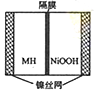 ��ͼ���ں����ø�ѹ������ػ����Ϸ�չ������һ�ֽ����⻯������أ�MH-Ni��أ��������й�˵���в���ȷ���ǣ�������
��ͼ���ں����ø�ѹ������ػ����Ϸ�չ������һ�ֽ����⻯������أ�MH-Ni��أ��������й�˵���в���ȷ���ǣ�������| A���ŵ�ʱ������ӦΪ��NiOOH+H2O+e-��Ni��OH��2+OH- |
| B����صĵ��Һ��ΪKOH��Һ |
| C�����ʱ������ӦΪ��MH+OH-��H2O+M+e- |
| D��MH��һ�ഢ����ϣ������ܶ�Խ��ص������ܶ�Խ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A�������£�0.2mol Fe������ˮ������Ӧ�����ɵ�H2������ĿΪ0.3NA | ||||
| B�������£�1L pH=13��NaOH��Һ�У���ˮ�����OH-������ĿΪ0.1NA | ||||
| C������ȼ�ϵ����������22.4L����״��������ʱ����·��ͨ���ĵ�����ĿΪ2NA | ||||
D��5NH4NO3
|
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com