
��֪XԪ��ԭ�ӵ�K��L��ĵ�����֮�ͱ�L��M��ĵ�����֮�Ͷ�1�����ӡ�YԪ
�ص�ԭ���������������ڲ��������3����ZԪ�غ�����3�����Ӳ㣬�������3�����ӡ�WԪ������ϼ�����ͻ��ϼ۾���ֵ��3������������������е���������Ϊ40%��
(1)Y��W����̬�⻯����ȶ���Ϊ(�û�ѧʽ��ʾ) �� ��
(2)X�����ڿ����м������ɵĻ������� ������(����ӡ����ۡ�)��
(3)X��Z������������Ӧˮ���ﷴӦ�����ӷ���ʽ ��
(4)W�ĵͼ���������Y���ʵ�ˮ��Һ��Ӧ�Ļ�ѧ����ʽ ��
(5)Y��Z�γɻ�����Ļ�ѧʽ�� ��ʵ���õ��˻����ﴦ�ڹ�̬��Һ̬ʱ�����磬����ˮ�ܵ��硣�ɴ��жϸû�������� ��(����ӡ����ۡ�)��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��15�֣���֪A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵�����������ӡ������Ϣ���±���ʾ�������ƶϻش��������⣺������ʱA��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ��
| A | A������������Ӧ��ˮ���ﻯѧʽΪH2AO3 |
| B | BԪ�صĵ�һ�����ܱ�ͬ������������Ԫ�ض��� |
| C | Cԭ����ͬ����ԭ���а뾶���ϡ��������⣩���䵥����ɫΪ��ɫ |
| D | Z�Ļ�̬ԭ�����������Ų�ʽΪ3s23p2 |
| E | E��Cλ�ڲ�ͬ���ڣ�Eԭ�Ӻ���������������C��ͬ�����������Ӿ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�±�ΪԪ�����ڱ���һ���֣�
| �� ���� | | | | |||||
| 1 | �� | | | | | | | |
| 2 | | | | | | �� | | |
| 3 | �� | | | �� | | �� | �� | |
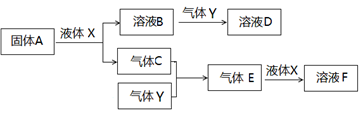
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���������ʰ���Ҫ���ɸߵ��͵�˳������
��1��NaF��NaCl�������ᡢSiC�������ʵ��۷е�˳�� ��
��2��C��N��O����Ԫ�صĵ�һ�����ܣ� ��
��3��H2O����Է���������H2SС�����е�ȴ�ߵ�ԭ�� ��
��4��ij��̬�Ŵط��ӵķ��ӽṹ��ͼ��ʾ���侧��ľ�����CO2����ľ���������ͬ������Ŵط��ӵķ���ʽΪ ��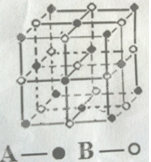
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ֶ�����Ԫ��A��B��C��D��E��ԭ��������������A��Cͬ�壬B��Dͬ�壬C���Ӻ�B���Ӿ�����ͬ�ĵ��Ӳ�ṹ��A��B��D��E�����γɹ��ۻ����A��B�γɵĻ�������ˮ�гʼ��ԣ�C��E�γɵĻ�������ˮ�г����ԡ�
�ش��������⣺
��1������Ԫ���У�ԭ�Ӱ뾶������ ���ǽ�������ǿ���� (��Ԫ�ط���)��
��2����A��B��D��E���γɵĹ����ͻ������У����ȶ��������� ���û�ѧʽ��ʾ����
��3��A��E�γɵĻ�������A��B�γɵĻ����ﷴӦ������Ļ�ѧʽΪ ,���д��ڵĻ�ѧ������Ϊ ��
��4������D�ڳ���ĵ���E��ȼ�գ���Ӧ�Ļ�ѧ����ʽΪ ��
��5������E��ˮ��Ӧ�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ֶ�����Ԫ��W��X��Y��Z��ԭ�����������������ϱ�����Ϣ�ش��������⡣
| | W | X | Y | Z |
| �ṹ������ | ����������Ӧ��ˮ����������̬�⻯�ﷴӦ�õ����ӻ����� | ��ɫ��Ӧ�ʻ�ɫ | ��ͬ��������Ԫ���γɵļ������У����Ӱ뾶��С | ��������������֮��Ϊ�� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������γɶ������ӣ���N3-��N3����NH2����NH4+��N2H5+��N2H62+�ȣ���֪N2H5+��N2H62+�������Է���X���H+�γɵģ���������NH4+�����ʡ�
�� 1��N3�����Ӻ��� �����ӣ�
�� �γ�N2H5+���ӵ����Է���X�ķ���ʽ�� ��
X�������п���ȼ�գ�д��ȼ�շ�Ӧ�Ļ�ѧ����ʽ ��
�� д��N2H62+������ǿ����Һ�з�Ӧ�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪X��Y��ZΪͬһ�����ڵ�����Ԫ�أ���ԭ�ӵIJ��ֵ�����(kJ��mol��1)���±���ʾ��
| | X | Y | Z |
| I1 | 496 | 738 | 578 |
| I2 | 4562 | 1451 | 1817 |
| I3 | 6912 | 7733 | 2745 |
| I4 | 9543 | 10540 | 11575 |
| ���� | NaCl | KCl | CaO |
| ������/(kJ��mol��1) | 786 | 715 | 3401 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪86Rn��뱣���̬ԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p63d104s24p64d104f145s25p65d106s26p6��88Ra��̬ԭ�ӵĵ����Ų�ʽ�ɼ�Ϊ[Rn]7s2��114��Ԫ���ǻ�ѧ�Һ�����ѧ�Һܸ���Ȥ����δ���ֵ�Ԫ�ء�
��1���ü���ʽд��114��Ԫ�ػ�̬ԭ�ӵĵ����Ų�ʽ�� ______________
________________________________________________________________��
��2������ԭ�Ӻ�������Ų����������ж�114��Ԫ�������ڱ��еĵ�________����________�塣
��3������114��Ԫ�������ڱ��е�λ���жϣ����Ӧ���е�������________��
��һ�����ܴ���88Ra
�ڱ��ֵ縺�Դ���3.0
������ϼ�Ϊ��4�ۣ�Ҳ���У�2��
�����������Ķ�Ӧˮ�����Լ���
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com