��2010?������ģ�⣩���Ϲ�����仯�����2009��12��7-18���ڸ籾�����ٿ����й�������ŵ����2020
�꣬��λGDP������̼�ŷű�2005���½�40%��45%��
��1����Ч����̼�����ֶ�֮һ�ǽ��ܣ��������ⷽ������ܵ���
C
C
��������ĸ��ţ�
A�����ˮ���⣺2H
2O
2H
2��+O
2�� B������ʹˮ�ֽ����⣺2H
2O
2H
2��+O
2��
C��̫������ֽ�ˮ���⣺2H
2O
2H
2��+O
2��
D����Ȼ�����⣺CH
4+H
2O
CO+3H
2��2��CO
2��ת�����л���ʵ��̼ѭ���������Ϊ1L���ܱ������У�����1mol CO
2��3mol H
2��һ�������·�����Ӧ��
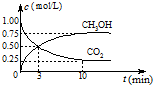
CO
2��g��+3H
2��g��?CH
3OH��g��+H
2O��g����H=-49.0kJ/mol�����CO
2��CH
3OH��g����Ũ����ʱ��仯��ͼ��ʾ��
�ٴ�3min��10min��v��H
2��=
0.11
0.11
mol/��L?min����
����˵��������Ӧ�ﵽƽ��״̬����
D
D
��ѡ���ţ���
A����Ӧ��CO
2��CH
3OH�����ʵ���Ũ��֮��Ϊ1��1����ͼ�н���㣩
B�����������ܶȲ���ʱ��ı仯���仯
C����λʱ����ÿ����3mol H
2��ͬʱ����1mol H
2O
D��CO
2����������ڻ�������б��ֲ���
�����д�ʩ����ʹn ��CH
3OH��/n ��CO
2���������
CD
CD
��ѡ���ţ���
A�������¶�B�����º��ݳ���He��g��
C����H
2O��g������ϵ�з��� D�����º����ٳ���1mol CO
2��3mol H
2����ͬ�¶��£����Ҫʹ������ƽ��Ũ��Ϊ1mol/L������ʼʱӦ�������г���1mol CO
2��
3.58
3.58
mol H
2��ƽ��ʱCO
2��ת����Ϊ
86.1%
86.1%
��
���ο����ݣ�
=2.64��
=21.166���������뱣��3λ��Ч���֣���
��3��CO
2����ϳ�DME�������ѣ��ǽ����ԴΣ�����о�����֮һ��
2CO
2��g��+6H
2��g����CH
3OCH
3��g��+3H
2O�������������ö���������ȼ�ϵ�أ�
��KOH��Һ���������Һ����д���õ�ع���ʱ������Ӧ�ĵ缫��Ӧ����ʽ
C2H6O+16OH--12e-�T2CO32-+11H2O
C2H6O+16OH--12e-�T2CO32-+11H2O
��




 CO+3H2
CO+3H2 CH3OH��g��+H2O��g����H=-49.0kJ/mol������ƽ�⣮
CH3OH��g��+H2O��g����H=-49.0kJ/mol������ƽ�⣮