
| c(NH4+)c(OH-) |
| c(NH3?H2O) |
| 10-13 |
| 10-11 |
| c(��)V(��)-c(��)V(��) |
| V(��)V(��) |
| c(NH4+)c(OH-) |
| c(NH3?H2O) |
| amol/L��10-7mol/L |
| bL��c(NH3?H2O)mol/L |
| a |
| 200b |
| a |
| 200b |
| 10-13 |
| 10-11 |
| 0.1b-0.01a |
| a+b |
| 0.05��2 |
| 2+9 |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ѡ�� | ��ijһ����������һ�� | ���� |
| A | FeSO4 ��NO2 ��MnO2��NaClO | H2SO3 |
| B | CH3COOH��HClO��H2S��HOOC-COOH | HF |
| C | �Ӿ۷�Ӧ�����ӷ�Ӧ�����ȷ�Ӧ��������ԭ��Ӧ | ��ɫ��Ӧ |
| D | ��NH4��2SO4��NH4Cl��NH4NO3��NH3?H2O | NH4HCO3 |
| A��A | B��B | C��C | D��D |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| A��пƬΪ�����������ܽ� |
| B��ͭƬΪ������ͭ����ʧ���Ӷ��ܵ����� |
| C��������пƬͨ����������ͭƬ��H+��ͭƬ�˶� |
| D����װ���ܽ�����ת��Ϊ��ѧ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A������ʯ��ˮ�������С�մ���Һ��Ӧ��Ca2++OH-+HCO3-��CaCO3��+H2O |
| B����NaAlO2��Һ��ͨ�����CO2��2AlO2-+CO2+3H2O��2Al��OH��3��+CO32- |
| C����NaHSO4��Һ�еμ�Ba��OH��2���պó�����ȫ��H++SO42-+Ba2++OH-��BaSO4��+H2O |
| D������0.1 mol���ʵ�FeI2��Һ��ͨ��0.1 mol��Cl2��2Fe+2I-+2Cl2��2Fe3++I2+4Cl- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
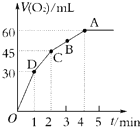 ����0.1mol MnO2��ĩ��50mL����������Һ��H2O2����=1.1g?mL-1���У��ڱ�״���·ų�����������ʱ��Ĺ�ϵ��ͼ��ʾ��
����0.1mol MnO2��ĩ��50mL����������Һ��H2O2����=1.1g?mL-1���У��ڱ�״���·ų�����������ʱ��Ĺ�ϵ��ͼ��ʾ��| 3 |
| 4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com