
�ⶨ�к���ʵ��װ����ͼ��
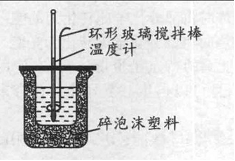
��1����ʵ����ϡ��Һ�У�H��(aq)��OH��(aq)��H2O(1)����H����57.3kJ/mol
������˵����ȷ���ǣ�_____________
A��ϡ�����ϡ NaOH��Һ��Ӧ���к���Ϊ -57.3kJ/mol
B��ϡ������ϡNaOH��Һ��Ӧ���к���Ϊ -57.3kJ/mol
C�� Ũ������ϡNaOH��Һ��Ӧ���к���Ϊ -57.3kJ/mol
D��ϡ������ϡNaOH��Һ��Ӧ����1molˮ���ų�57.3kJ����
������NaOH��Һ����Ba(OH)2��Һ�����ⶨ��������ϡ������Һ�м���100mL0.4mol/L Ba(OH)2��Һ���ų�����5.12kJ����ͨ�����������ʵ���е����к��ȱ仯��ԭ��______________
(2)ȡ50mL0.5mol/LNaOH��Һ��30mL0.5mol/Lϡ�������ʵ�飬������±���ʾ��
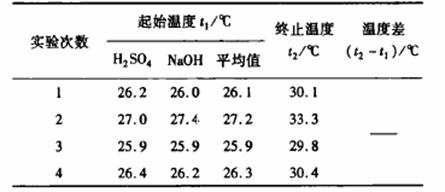
���������Ϊ������Һ���ܶȶ���1g/cm3���кͺ����ɵ���Һ������c=4.18J/(g��oC)�����к��� H=__________kJ/mol��С�������1λ��������ʵ��������ֵ��57.3 kJ/mol ��ƫ��,����ƫ���ԭ�������(����ĸ)________________
H=__________kJ/mol��С�������1λ��������ʵ��������ֵ��57.3 kJ/mol ��ƫ��,����ƫ���ԭ�������(����ĸ)________________
A .ʵ��װ�ñ��¸���Ч����
B .����ȡNa0H ��Һ�����ʱ���Ӷ���
C .�ֶ�ΰ�NaOH ��Һ����ʢ�������С�ձ���
D .���¶ȼƲⶨNa0H ��Һ����ʼ�¶Ⱥ�ֱ�ӲⶨH2S04 ��Һ���¶�

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����������������������ϡ���ᷴӦ���ų�NO���ʵ���������
A��FeO B. Fe2O3 C. FeSO4 D��Fe3O4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij�¶�ʱ��Ũ�ȶ�Ϊ1mol/L����������X2��Y2�����ܱ������з�Ӧ��������Z��4s��X2��Y2��Z��Ũ�ȷֱ���0.4mol/L��0.8mol/L��0.4mol/L����Z��X��Y��ʾ����û�ѧ����ʽ�ɱ�ʾΪ( )��
A��X2+2Y2 ==2XY2 B��2X2+Y2 ==X2Y C��3X2+Y2 ==2X3Y D��X2+3Y2 ==2XY3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����йػ�ѧ�о�����ȷ˵����
A��ͬʱ�ı������������о���Ӧ���ʵı仯���ܸ���ó��йع���
B������ͬһ����ѧ��Ӧ������������Ӧ�����淴Ӧ���䷴Ӧ���ʱ���ͬ
C������11-17��Ԫ������������Ӧˮ���������Եݱ���ɣ��Ƴ�ͬ����Ԫ�صĽ����ԡ�
�ǽ����Եݱ����
D����HF��HCl��HBr��HI���Ե�������ʵ���Ƴ�F��Cl��Br��I�ķǽ��������Ĺ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��(H2NNH2)��һ�ָ���ȼ�ϣ��йػ�ѧ��Ӧ�������仯��ͼ��ʾ����֪����1mol��ѧ�������������kJ����N��NΪ942��O=OΪ500��N-NΪ154�������1molN-H�������������KJ����
A.194 B.391 C.526.7 D.658

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���������ʣ��ټ��� �ڱ� �۾���ϩ ����Ȳ ��1����ϩ ������ �ױ������м���ʹ���Ը��������Һ��ɫ������ʹ��ˮ������Ӧ����ɫ���ǣ� ��
A���٢ܢ� B���ܢ� C���ܢݢ� D���ۢܢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���з�Ӧ����ȡ����Ӧ���� (����)��
��CH3CH===CH2��Br2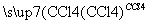 CH3CHBrCH2Br
CH3CHBrCH2Br
��CH3CH2OH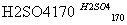 CH2===CH2����H2O
CH2===CH2����H2O
��CH3COOH��CH3CH2OH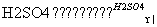 CH3COOCH2CH3��H2O
CH3COOCH2CH3��H2O
��C6H6��HNO3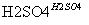 C
C 6H5NO2��H2O
6H5NO2��H2O
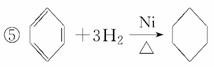
��2CH3CH2OH��O2 2CH3CHO��2H2O
2CH3CHO��2H2O
����ά������ˮ������������
����֬��������Ӧ
�ᰱ�������ɶ��ĵķ�Ӧ
��nCH2===CH2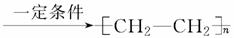
A���ۢܢߢ� B���٢ڢۢ�
B���٢ڢۢ�
C���ݢޢߢ� D���ۢܢޢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���й����л��������˵����ȷ����
A�������������������Na2CO3��Һ�������� B.���飨C5H12��������ͬ���칹��
C.��ϩ��������ϩ�ͱ����Ӿ�����̼̼˫�� D.���ࡢ��֬�͵����ʾ��ɷ���ˮ�ⷴӦ
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com