
�ֶ�0.1 mol/L�Ĵ�����Һ��������о���
��1����pH��ֽ�ⶨ����Һ��pH������ȷ�IJ���
��
��2��������Һ�ʼ��Ե�ԭ����(�����ӷ���ʽ��ʾ) ��
��3��ijͬѧ��Ϊ����Һ��Na2CO3��ˮ�������ģ�����ˮ���CO32�����Ӳ�������������10�����������ʵ��֤����ͬѧ�Ĺ۵��Ƿ���ȷ��
��
��4��ijͬѧ������ѧ֪ʶ����Һ���з� ����д���������ĸ���ϵʽ�������жϣ�����ȷ�Ĺ�ϵʽ�����
����д���������ĸ���ϵʽ�������жϣ�����ȷ�Ĺ�ϵʽ����� �����ڴ���ĺ���д����ȷ�Ĺ�ϵʽ��
�����ڴ���ĺ���д����ȷ�Ĺ�ϵʽ��
�� c()�� 2[c(2��3 )��c(HCO��3 ) ]�� ��
�� c()��c(H+)�� 2 c(2��3 )��c(HCO��3 )��c(OH��)�� ��
�� c(OH��)�� c(H+)��c(HCO��3 ) ��c()�� ��
�� c()��c(2��3 )��c(OH��)��c(HCO��3 ) �� ��
��1����С��pH��ֽ���ڱ�������Ƭ���ϣ��ò�����պȡ����Һ������ֽ���в�������ֽ��ɫ�������ɫ���Ա�ȷ����Һ��pH ֵ(2��)
��2��CO32��+H2O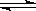 HCO3��+OH��(2��)
HCO3��+OH��(2��)
��3����pH��ֽ����pH�ƣ��ⳣ����0.1 mol/L������Һ��pH����pH��12�����ͬѧ�Ĺ۵���ȷ����pH��12�����ͬѧ�Ĺ۵㲻��ȷ��(2��)
��4����c() ="== " 2[c(2��3 )��c(HCO��3 ) ��c()] (1��) �� (1��)
(1��)
��c(OH��) === c(H+)��c(HCO��3 ) ��2c() (1��) �� (1��)
(1��)
����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ֶ�0.1 mol/L�Ĵ�����Һ��������о���
��1����pH��ֽ�ⶨ����Һ��pH������ȷ�IJ���
��
��2��������Һ�ʼ��Ե�ԭ����(�����ӷ���ʽ��ʾ) ��
��3��ijͬѧ��Ϊ����Һ��Na2CO3��ˮ�������ģ�����ˮ���CO32�����Ӳ�������������10�����������ʵ��֤����ͬѧ�Ĺ۵��Ƿ���ȷ��
��
��4��ijͬѧ������ѧ֪ʶ����Һ���з�����д���������ĸ���ϵʽ�������жϣ�����ȷ�Ĺ�ϵʽ�����![]() �����ڴ���ĺ���д����ȷ�Ĺ�ϵʽ��
�����ڴ���ĺ���д����ȷ�Ĺ�ϵʽ��
�� c()�� 2[c(2��3 )��c(HCO��3 ) ]�� ��
�� c()��c(H+)�� 2 c(2��3 )��c(HCO��3 )��c(OH��)�� ��
�� c(OH��)�� c(H+)��c(HCO��3 ) ��c()�� ��
�� c()��c(2��3 )��c(OH��)��c(HCO��3 ) �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ֶ�0.1 mol/L�Ĵ�����Һ��������о���
��1����pH��ֽ�ⶨ����Һ��pH������ȷ�IJ���
��
��2��������Һ�ʼ��Ե�ԭ����(�����ӷ���ʽ��ʾ) ��
��3��ijͬѧ��Ϊ����Һ��Na2CO3��ˮ�������ģ�����ˮ���CO32�����Ӳ�������������10�����������ʵ��֤����ͬѧ�Ĺ۵��Ƿ���ȷ��
��
��4��ijͬѧ������ѧ֪ʶ����Һ���з�����д���������ĸ���ϵʽ�������жϣ�����ȷ�Ĺ�ϵʽ����������ڴ���ĺ���д����ȷ�Ĺ�ϵʽ��
�� c()�� 2[c(2��3 )��c(HCO��3 ) ]�� ��
�� c()��c(H+)�� 2 c(2��3 )��c(HCO��3 )��c(OH��)�� ��
�� c(OH��)�� c(H+)��c(HCO��3 ) ��c()�� ��
�� c()��c(2��3 )��c(OH��)��c(HCO��3 ) �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�����ʡ�人�и߶���ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ������
�ֶ�0.1 mol/L�Ĵ�����Һ��������о���
��1����pH��ֽ�ⶨ����Һ��pH������ȷ�IJ��� ��
��2�����ϲ�ô�����Һ�ʼ��ԣ��������ӷ���ʽ��ʾ��ԭ�� ��
��3��ijͬѧ������ѧ֪ʶ����Һ���з�����д���������ĸ���ϵʽ�������жϣ�����ȷ�Ĺ�ϵʽ����� �����ڴ���ĺ���д����ȷ�Ĺ�ϵʽ��
�����ڴ���ĺ���д����ȷ�Ĺ�ϵʽ��
�� c()�� 2[c(2��3 )��c(HCO��3 ) ]�� ��
�� c()��c(H+)�� 2 c(2��3 )��c(HCO��3 )��c(OH��)�� ��
�� c(OH��)�� c(H+)��c(HCO��3 ) ��c()�� ��
�� c()��c(2��3 )��c(OH��)��c(HCO��3 ) �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012��ӱ�ʡ�е�����У�߶���ѧ����ĩ������ѧ�Ծ� ���ͣ������
�ֶ�0.1 mol/L�Ĵ�����Һ��������о���
��1����pH��ֽ�ⶨ����Һ��pH������ȷ�IJ���
��
��2��������Һ�ʼ��Ե�ԭ����(�����ӷ���ʽ��ʾ) ��
��3��ijͬѧ��Ϊ����Һ��Na2CO3��ˮ�������ģ�����ˮ���CO32�����Ӳ�������������10�����������ʵ��֤����ͬѧ�Ĺ۵��Ƿ���ȷ��
��
��4��ijͬѧ������ѧ֪ʶ����Һ���з�����д���������ĸ���ϵʽ�������жϣ�����ȷ�Ĺ�ϵʽ����� �����ڴ���ĺ���д����ȷ�Ĺ�ϵʽ��
�����ڴ���ĺ���д����ȷ�Ĺ�ϵʽ��
�� c()�� 2[c(2��3 )��c(HCO��3 ) ]�� ��
�� c()��c(H+)�� 2 c(2��3 )��c(HCO��3 )��c(OH��)�� ��
�� c(OH��)�� c(H+)��c(HCO��3 ) ��c()�� ��
�� c()��c(2��3 )��c(OH��)��c(HCO��3 ) �� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com