

 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
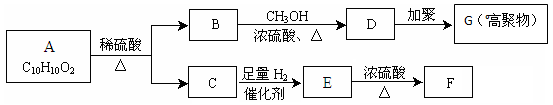
 ��F��ʹ���CCl4��Һ��ɫ����ش��������⣺
��F��ʹ���CCl4��Һ��ɫ����ش��������⣺�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
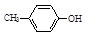
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
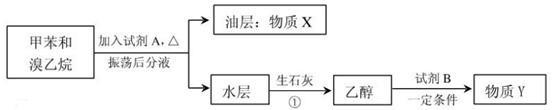
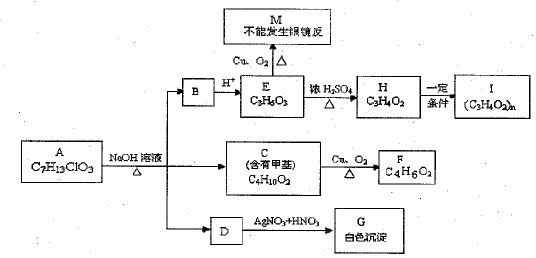
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
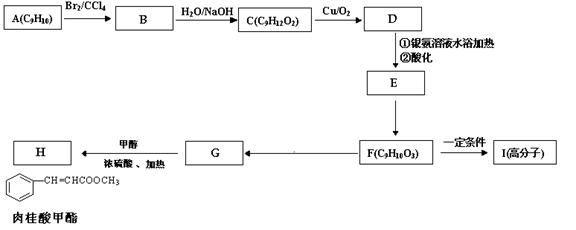
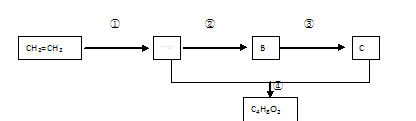
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
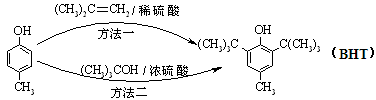
A��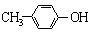 ����Na2CO3��Һ��Ӧ ����Na2CO3��Һ��Ӧ |
B�� ��BHT��Ϊͬϵ�� ��BHT��Ϊͬϵ�� |
| C��BHT�����ڿ����лᱻ���� |
| D�����ַ����ķ�Ӧ���Ͷ��Ǽӳɷ�Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
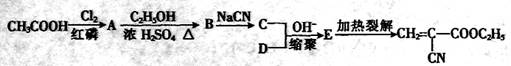

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
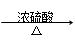 (i) ��25��ʱ������ƽ�ⳣ��K1��3.9��10��4��K2��5.5��10��6
(i) ��25��ʱ������ƽ�ⳣ��K1��3.9��10��4��K2��5.5��10��6 (ii) A + RCOOH(��ROH)
(ii) A + RCOOH(��ROH) ����ζ�IJ���
����ζ�IJ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
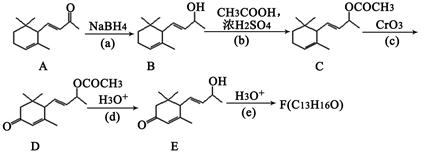
| A�����������ڷ����廯���� | B���÷�������4�ֲ�ͬ���͵���ԭ�� |
| C��1mol��������������2molNaOH | D���������ܷ���������Ӧ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com