
��18�֣�ÿ��3�֣�Ǧ�����ǵ��͵Ŀɳ��͵�أ���������������Ƕ��Բ��ϣ�����ܷ�ӦʽΪ��Pb+PbO2+4H++2SO42����![]() 2PbSO4+2H2O
2PbSO4+2H2O
��ش��������⣨�������⡢����������ԭ����
��1���ŵ�ʱ�������ĵ缫��Ӧʽ��______ ______�����Һ��H2SO4��Ũ�Ƚ���__ __�������·ͨ��1 mol����ʱ�������ϸ��������������___ ___g��
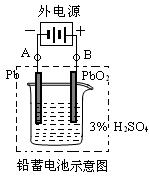
��2�����ʹ��һ��ʱ�������ͼ���ӣ�����A�缫������___ __��B�缫������__ __����ʱǦ���ص��������ļ��Խ�_ ___��������䡱�Ե�����
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ʵ���⣺(18�֣�ÿ��2��)
�֣�
�ס��ҡ�����λͬѧ�ֱ�������ʵ��װ�ü���ѧҩƷ����ʯ��Ϊ�������ƺ���ʯ�ҵĻ�����ȡ�������������̽�������ش����⣺
��1��������ȡ�����Ļ�ѧ����ʽΪ�� ��
��2����λͬѧ���������ſ������ռ���������ԭ���� ��
��3����λͬѧ������װ����ȡ����ʱ,������һλͬѧû���ռ�������ʵ���������ȷ��������Ϊû���ռ���������ͬѧ��___��(���ס������ҡ�����)��
��4�����鰱���Ƿ��ռ����ķ�����:��������������������ͽ��ۣ�
��5����λͬѧ����Ϊ���ǵ�ʵ��װ�û������ڼ���̼����粒�������ȡ�����İ��������ж��ܹ��ﵽʵ��Ŀ�ĵ���________(��ס������ҡ�����)����װ���е�NH4HCO3�����ܷ���NH4Cl������棿____ (��ܡ����ܡ�)��
��
��֪���������ʹ���������Һ��ɫ��Ӧ�Ļ�ѧ����ʽΪ��
5SO2+2KMnO4+2H2O=K2SO4+2MnSO4+2H2SO4
��ͼΪ��֤Ũ������ľ̿�ڼ��������£���Ӧ�������Ƿ���SO2��CO2�IJ���װ�á�

��6��ʵ��ʱ���� ������ʢ�г���ʯ��ˮ��ʵ��װ��(��a��b ���)��
��7���ɹ۲쵽Aƿ��Һ ��
��8��Cƿ��Һ�������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010��2011�꽭���γ���ѧ�߶���ѧ����ĩ���Ի�ѧ����ѡ�ޣ� ���ͣ������
��18�֣�ÿ��3�֣�Ǧ�����ǵ��͵Ŀɳ��͵�أ���������������Ƕ��Բ��ϣ�����ܷ�ӦʽΪ��Pb+PbO2+4H++2SO42���� 2PbSO4+2H2O
2PbSO4+2H2O
��ش��������⣨�������⡢����������ԭ����
��1���ŵ�ʱ�������ĵ缫��Ӧʽ��______ ______�����Һ��H2SO4��Ũ�Ƚ���__ __�������·ͨ��1 mol����ʱ�������ϸ��������������___ ___g��
��2�����ʹ��һ��ʱ�������ͼ���ӣ�����A�缫������___ __��B�缫������__ __����ʱǦ���ص��������ļ��Խ�_ ___��������䡱�Ե�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011��ɽ��ʡ���϶��и�һ��һѧ��������⻯ѧ�Ծ� ���ͣ�ʵ����
(18�֣�ÿ��2��)
�֣�
�ס��ҡ�����λͬѧ�ֱ�������ʵ��װ�ü���ѧҩƷ����ʯ��Ϊ�������ƺ���ʯ�ҵĻ�����ȡ�������������̽�������ش����⣺
��1��������ȡ�����Ļ�ѧ����ʽΪ��_____________ _________ _______��
��2����λͬѧ���������ſ������ռ���������ԭ����_________ _________��
��3����λͬѧ������װ����ȡ����ʱ,������һλͬѧû���ռ�������ʵ���������ȷ��������Ϊû���ռ���������ͬѧ��___ ___��(���ס������ҡ�����)��
��4�����鰱���Ƿ��ռ����ķ�����:��������������������ͽ��ۣ�
_______ _____��
��5����λͬѧ����Ϊ���ǵ�ʵ��װ�û������ڼ���̼����粒�������ȡ�����İ��������ж��ܹ��ﵽʵ��Ŀ�ĵ���________(��ס������ҡ�����)����װ���е�NH4HCO3�����ܷ���NH4Cl������棿____ _(��ܡ����ܡ�)��
��
��֪���������ʹ���������Һ��ɫ��Ӧ�Ļ�ѧ����ʽΪ��
5SO2+2KMnO4+2H2O=K2SO4+2MnSO4+2H2SO4
��ͼΪ��֤Ũ������ľ̿�ڼ��������£���Ӧ�������Ƿ���SO2��CO2�IJ���װ�á�
��6��ʵ��ʱ���� ������ʢ�г���ʯ��ˮ��ʵ��װ��(��a��b ���)��
��7���ɹ۲쵽Aƿ����Һ ��
��8��Cƿ��Һ�������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011��ɽ��ʡ��һ��һѧ��������⻯ѧ�Ծ� ���ͣ�ʵ����
(18�֣�ÿ��2��)
�֣�
�ס��ҡ�����λͬѧ�ֱ�������ʵ��װ�ü���ѧҩƷ����ʯ��Ϊ�������ƺ���ʯ�ҵĻ�����ȡ�������������̽�������ش����⣺
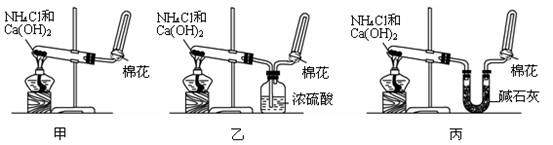
��1��������ȡ�����Ļ�ѧ����ʽΪ��_____________ _________ _______��
��2����λͬѧ���������ſ������ռ���������ԭ����_____ ____ _________��
��3����λͬѧ������װ����ȡ����ʱ,������һλͬѧû���ռ�������ʵ���������ȷ��������Ϊû���ռ���������ͬѧ��___ ___��(���ס������ҡ�����)��
��4�����鰱���Ƿ��ռ����ķ�����:��������������������ͽ��ۣ�
_______ _____��
��5����λͬѧ����Ϊ���ǵ�ʵ��װ�û������ڼ���̼����粒�������ȡ�����İ��������ж��ܹ��ﵽʵ��Ŀ�ĵ���________(��ס������ҡ�����)����װ���е�NH4HCO3�����ܷ���NH4Cl������棿____ _(��ܡ����ܡ�)��
��
��֪���������ʹ���������Һ��ɫ��Ӧ�Ļ�ѧ����ʽΪ��
5SO2+2KMnO4+2H2O=K2SO4+2MnSO4+2H2SO4
��ͼΪ��֤Ũ������ľ̿�ڼ��������£���Ӧ�������Ƿ���SO2��CO2�IJ���װ�á�
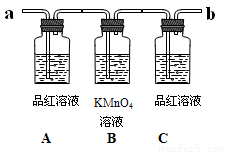
��6��ʵ��ʱ���� ������ʢ�г���ʯ��ˮ��ʵ��װ��(��a��b ���)��
��7���ɹ۲쵽Aƿ����Һ ��
��8��Cƿ��Һ�������� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com