
�����������ض�����Ҫ�Ĺ�ҵ��Ʒ����ش�:
(1)��ҵұ�����Ļ�ѧ����ʽ������������������������
(2)��������������Һ��Ӧ�����ӷ���ʽ��������������������������������
(3)��ҵƷ�������ص���Һ�к���ijЩ����������,�������ӽ���Ĥ������ᴿ��
������װ�������ӽ���Ĥ(ֻ����������ͨ��),�乤��ԭ����ͼ��ʾ��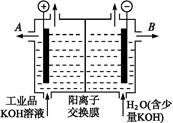
�ٸõ��۵�������Ӧʽ������������������������
��ͨ�翪ʼ��,����������ҺpH������,�����ԭ��������������������������
�۳�ȥ���ʺ������������Һ����Һ������������(��д��A����B��)������

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��8�֣���������ȵ���Ƭ��ͭƬ�õ�����������500mL����ͭ��Һ�й�����ͼ1��װ�ã�
�����¾����跴Ӧ��������Һ������䣩��
����Ƭ�ϵĵ缫��ӦʽΪ ��
��ͭƬ��Χ��Һ����� ������
����2 min������Ƭ��ͭƬ֮���������Ϊ1��2g�����㣺
�ٵ����������ĵ��ӵ����ʵ���Ϊ mo1��
�ڸö�ʱ����������ͭ��ʾ��ƽ����Ӧ����Ϊ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ����ʯīΪ�缫���CuCl2��Һ��װ��ʾ��ͼ��
��1��������������ת������Ҫ��ʽ�� ��
��2�����һ��ʱ���b�缫�Ͽɹ۲쵽�������� ��
��3���������У�a�缫Ϊ �����õ缫�Ϸ����ĵ缫��ӦʽΪ�� ������õ缫����ķ��� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�绯ѧԭ���ڹ�ҵ������������Ҫ�����ã���������ѧ֪ʶ�ش��й����⡣
(1)�õ��ķ�����������Һ����Ϊ��������о�������Ҫ��ʵ�����壬������ת��Ϊ�������ǵ�ⷨ�������������һ����Ҫ���ݡ����ǵ������������ʵ��װ�ã�
����֪�����ķ�ӦΪ(x��1)S2��=Sx��S2����2xe�����������ĵ缫��Ӧʽ��____________________________
����Ӧת��x mol����ʱ���������������Ϊ____________(��״����)��
�ڽ�Na2S��9H2O����ˮ������������Һʱ��ͨ�����ڵ����������ܽ⡣��ԭ����(�����ӷ�Ӧ����ʽ��ʾ)��___________________________��
(2)MnO2��һ����Ҫ�������ܲ��ϣ��Ʊ�MnO2�ķ���֮һ����ʯīΪ�缫������ữ��MnSO4��Һ�������ĵ缫��ӦʽΪ______________________������Ǧ����Ϊ��Դ����ữ��MnSO4��Һ����ͼ��ʾ��Ǧ���ص��ܷ�Ӧ����ʽΪ_______________________��
����������4 mol H��������ʱ�����·��ͨ���ĵ��ӵ����ʵ���Ϊ________��MnO2�����۲���Ϊ________g��
(3)��ͼ���װ�ÿ��Ƶþ��о�ˮ���õ� ��ʵ������У������������������Y������Һ������
��ʵ������У������������������Y������Һ������

�ٵ������У�X������Һ��pH________(�������С�����䡱)��
�ڵ������У�Y�������ĵ缫��ӦΪFe��6e����8OH��= ��4H2O��______________________________������X���ռ���672 mL���壬��Y���ռ���168 mL����(��������Ϊ��״��ʱ�������)����Y�缫(���缫)��������________g��
��4H2O��______________________________������X���ռ���672 mL���壬��Y���ռ���168 mL����(��������Ϊ��״��ʱ�������)����Y�缫(���缫)��������________g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ȤС��Ϊ����ߵ�ص�Ч��,�������ͼ��ʾ��ԭ��ء�
��ش���������:
(1)��X��AlCl3��Һ,Y��ϡ����,����д���缫���Ƽ��缫��Ӧ:
AlƬ(����)��____________________________,
CuƬ(����)��____________________________��
(2)��X��Ũ����,Y��NaCl��Һ,����д���缫���Ƽ��缫��Ӧ:
AlƬ(����)��____________________________,
CuƬ(����)��____________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ��ʾ���ڲ�ͬ�ĵ������Һ�п�����ɲ�ͬ�ĵ�ء�
(1)�ٵ��������ҺΪϡ����ʱ��Fe�缫�� (���������)������缫��ӦʽΪ
��
�ڵ��������ҺΪNaOH��Һʱ��Al�缫�� (���������)������缫��ӦʽΪ ��
(2)��������Ϊп���������ҺΪŨ���ᣬ��Fe�缫�� (���������)������缫��ӦʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼΪ������ļ����������أ�X��YΪֱ����Դ�������缫���������У��� ��ʯī�缫�����ȱ�졣��ش�
(1)��ԴX��Ϊ ��(���������)���ҳ���Pt�缫�ϵĵ缫��ӦʽΪ ��
(2)�׳���Ϊ��⾫��ͭ��װ�ã�����������12.8 g�����ҳ��������Ϸų��������ڱ�״���µ����Ϊ ����·��ͨ���ĵ���Ϊ mol��
(3)��(2)������£����ҳ�ʣ����Һ�������Ϊ400 mL�������������Һc(OH��)�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijԭ���װ����ʢ��ŨKOH��Һ�����ֱ���__________(�������������ͬ)��ͨ���ȼ�����壬��__________��ͨ��O2�����·�о��е���ͨ��������������__________������__________������Һ��OH����__________���ƶ���������������⣺
| | ��ȼ������ | ������Ӧ | ������Ӧ | ����ܷ�Ӧ | pH�仯 |
| (1) | H2 | | | | |
| (2) | CO | | | | |
| (3) | CH4 | | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�缫a��b�ֱ�ΪAg�缫��Pt�缫���缫c�� d�� �� ʯī�缫��ͨ��һ��ʱ�����c��d�����Ϲ��ռ���336mL����״̬�����塣�ش�
��1��ֱ����Դ�У�MΪ______________����
��2��Pt�缫�����ɵ�������____________��������Ϊ____________g ��
��3������ת�Ƶ����ʵ�����缫b��c��d�ֱ����ɵ����ʵ����ʵ���֮��Ϊ________��
��4���������У�AgNO3��Һ��Ũ��______________�����������С�����䡱��
��ͬ����AgNO3��Һ��pH__________��H2SO4��Һ��Ũ��_________ ��H2SO4��Һ��pH________ ��
��5����H2SO4��Һ������������5��00%��Ϊ5��02%����ԭ��5��00%��H2SO4��ҺΪ______g��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com