
���ж���Ӧ�л����������ȫ��ȷ����(����)
�ټ��飺��Ȼ������Ҫ�ɷ֣��ܷ���ȡ����Ӧ������ϩ������һ������ʯ�ͻ�����չˮƽ�ı�־�����Է����ӳɷ�Ӧ���۱���ƽ��ṹ��ÿ�������к���3��̼̼˫��
����֬�����ڸ߷��ӻ�������Է���ˮ�ⷴӦ���ݵ��ۣ������������ʣ�����Ԫ�ر���ɫ�������ʣ�ˮ������ղ���Ϊ�����ᣬ�������ĵ�������Ũ�����Ի�ɫ
A���٢ڢ� B���ڢܢ�
C���٢ݢ� D���٢ڢ�
 Ӣ��СӢ������Ĭдϵ�д�
Ӣ��СӢ������Ĭдϵ�д� �����ҵ���������ͯ������ϵ�д�
�����ҵ���������ͯ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������л�ԭ�ԣ����Ժ�������������Ӧ�������������£�H2S��KMnO4��Ӧ����S��MnSO4��K2SO4��H2O��д���÷�Ӧ�Ļ�ѧ����ʽ��_________________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������������Һ���ܴ��������һ����
A��Fe3+��H+��ClO-��SO32- B��K+��Al3+��Cl-��CO32-
C��Na+��Ca2+��HCO3-��OH- D��Fe2����NH4+��NO3����SO42-
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
̼������� ��DMC)��һ�����͵���ɫ�л��ϳ��м��壬���������о���ʹ�ð�ȫ�����㡢��Ⱦ�١�����������ص㡣��֪һ�������£�̼��������ܷ������·�Ӧ��
��DMC)��һ�����͵���ɫ�л��ϳ��м��壬���������о���ʹ�ð�ȫ�����㡢��Ⱦ�١�����������ص㡣��֪һ�������£�̼��������ܷ������·�Ӧ��
��Ӧ�٣�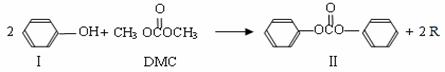
��Ӧ�ڣ�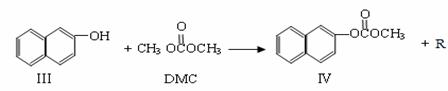
��1���������ķ���ʽΪ  ����Ӧ�ٺͷ�Ӧ����R�Ľṹ��ʽΪ ��
����Ӧ�ٺͷ�Ӧ����R�Ľṹ��ʽΪ ��
��2�� DMC������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ ��
DMC������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ ��
��3�������ڷ�Ӧ�٣�DMC�����ᱽ����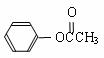 ���ڴ���������Ҳ�����ɻ������ͬʱ�õ�һ�ָ���ƷG�������й�G��˵����ȷ���� ��
���ڴ���������Ҳ�����ɻ������ͬʱ�õ�һ�ָ���ƷG�������й�G��˵����ȷ���� ��
A��G�������Ǽ�������
B�������G��Ϊͬ���칹��
C��һ�������£�G�ܷ���ˮ�ⷴӦ
D��G��������Cu(OH)2��Ӧ���ɺ�ɫ����
��4����Ӧ���з�Ӧ��������ͬ���ɵõ�������H��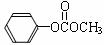 ����H��ͬ���칹���ж��֣���д����������������H��һ��ͬ���칹�� ��
����H��ͬ���칹���ж��֣���д����������������H��һ��ͬ���칹�� ��
A����FeCl3��Һ����ɫ��B�������ϵĺ˴Ź�������������壬C���ܷ���������Ӧ
��5����̼��������ϳ�ɱ�����ά���·�����£�
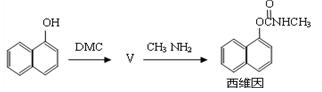
������������ķ�Ӧ�����ڷ�Ӧ�ڣ�����Ľṹ��ʽΪ ��1mol��ά�������� mol H2�����ӳɷ�Ӧ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
a mL������̬���Ļ������������������ϵ�ȼ��ը�ָ���ԭ����״̬(���¡���ѹ)���������С2a mL����������������(����)
A��CH4��C2H4��C3H4 B��C2H6��C3H6��C4H6
C��CH4��C2H6��C3H8 D��C2H4��C2H2��CH4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����������ȷ���ǣ� ��
A��ij�л���ȼ�պ����ֻ��CO2��H2O�����Ƴ��Ľ������л���������
B��ij�л���ȼ�պ����ֻ��CO2��H2O�����Ƴ��Ľ������л����������ĺ���������
C��ij�л���ȼ�պ����ֻ��CO2��H2O��ͨ���ⶨ�л��CO2��H2O����������ȷ��
�л����Ƿ�����
D��������ͬϵ���к�̼����ߣ�����������Դ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������A�����ϣ��������к�C 72.0%��H 6.67%������Ϊ��������������֪A����Է�������Ϊ150���ִ����������л�������ķ��ӽṹ���������ַ�����
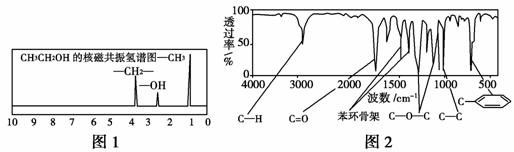
����һ���˴Ź����ǿ��Բⶨ�л�������ﲻͬ��ѧ��������ԭ�Ӽ���������������Ҵ�(CH3CH2OH)�ĺ˴Ź���������3���壬�����֮��Ϊ3��2��1����ͼ1��ʾ���ֲ��A�ĺ˴Ź���������5���壬�����֮��Ϊ1��2��2��2��3��
�����������ú�������ǿɳ�������л��������е�ijЩ���ţ��ֲ��A���ӵĺ��������ͼ2��ʾ��
��֪��A������ֻ��һ���������ұ�����ֻ��һ��ȡ�������Իش��������⣺
(1)A�ķ���ʽΪ��_______________________________________________��
(2)A�Ľṹ��ʽΪ��______________________________________________��
(3)A�ķ�����ͬ���칹���ж��֣��밴Ҫ��д���������ֽṹ��ʽ��
�ٷ����в������ķ����______________________________________��
�ں��ǻ��ұ�����ֻ������ȡ�����ķ���ȩ��________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���������缫������KOH��Һ�У�Ȼ��ֱ�������ͨ��CH4��O2�����ɲ�������������������ȷ���� (����)
��ͨ��CH4�ĵ缫Ϊ�������������ĵ缫��ӦʽΪO2��2H2O��4e��===4OH������ͨ��CH4�ĵ缫��ӦʽΪCH4��2O2��4e��===CO2��2H2O���ܸ����ĵ缫��ӦʽΪCH4��10OH����8e��===CO ��7H2O���ݷŵ�ʱ��Һ�е����������ƶ����ŵ�ʱ��Һ�е����������ƶ�
��7H2O���ݷŵ�ʱ��Һ�е����������ƶ����ŵ�ʱ��Һ�е����������ƶ�
A���٢ۢ� B���ڢܢ�
C���ܢݢ� D���٢ڢ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com