



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ˮԡ |
| ˮԡ |
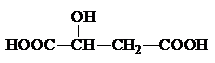 ���÷����й����ŵ�����Ϊ
���÷����й����ŵ�����Ϊ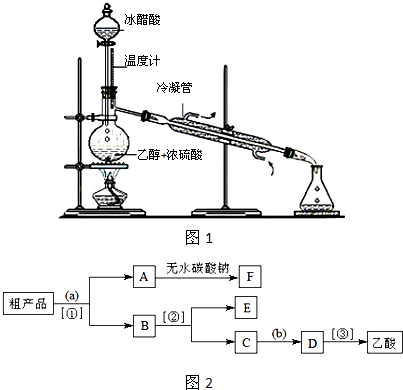
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 �л���ѧ֪ʶ��������Ӧ�ù㷺��
�л���ѧ֪ʶ��������Ӧ�ù㷺���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ÿ100g���� Ӫ���ɷ� |
���� | ��֬ | ������ | ά����C | �� | þ | �� | п |
| 7.6g | 7.8g | 7.4g | 18mg | 201mg | 18.2mg | 30.8mg | 8.1mg |
| ����� | 1 | 2 | 3 |
| ����ʱ����ζ | ������ζ | ��ֽ��ζ | �ս���ë��ζ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(8��)��1�����ࡢ��֬�������ʶ�����������Ӫ�����ʡ�
����֬�������������ø��������ˮ��Ϊ��֬����� ��д���ƣ���
�ڰ���������ɵ����ʵĻ����ṹ��Ԫ���������һ�����еĹ������ǰ�������NH2����
` ��д���ƣ���
�۵����ڵ���ø������������ˮ��Ϊ�����ǣ�C6H12O6�������������������ڱ��������ɶ�����̼��ˮ��д�������������ڱ������Ļ�ѧ����ʽ�� ��
��2����������һ�������˿ڵĽ�����衣���������ijɷ��ж��֣����ᱽ���������е�һ�֣������Դ���������ȡ��Ҳ�����üױ����Ҵ�Ϊԭ�Ͻ����˹��ϳɡ�һ�ֺϳ�·�����£�
����C�Ľṹ��ʽΪ ��
����д����Ӧ�ٵĻ�ѧ����ʽ�� ��
������Ӧ�۵ķ�Ӧ����Ϊ ��
������Ӧ ������ţ�ԭ�ӵ�����������Ϊ100����������ɫ��ѧ��Ҫ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�꽭��ʡ���Ƹ���ѧ��һ�ڶ�ѧ����ĩ���Ի�ѧ���� ���ͣ������
(8��)��1�����ࡢ��֬�������ʶ�����������Ӫ�����ʡ�
����֬�������������ø��������ˮ��Ϊ��֬����� ��д���ƣ���
�ڰ���������ɵ����ʵĻ����ṹ��Ԫ���������һ�����еĹ������ǰ�������NH2����
` ��д���ƣ���
�۵����ڵ���ø������������ˮ��Ϊ�����ǣ�C6H12O6�������������������ڱ��������ɶ�����̼��ˮ��д�������������ڱ������Ļ�ѧ����ʽ�� ��
��2����������һ�������˿ڵĽ�����衣���������ijɷ��ж��֣����ᱽ���������е�һ�֣������Դ���������ȡ��Ҳ�����üױ����Ҵ�Ϊԭ�Ͻ����˹��ϳɡ�һ�ֺϳ�·�����£�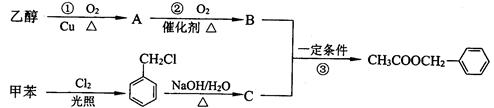
����C�Ľṹ��ʽΪ ��
����д����Ӧ�ٵĻ�ѧ����ʽ�� ��
������Ӧ�۵ķ�Ӧ����Ϊ ��
������Ӧ ������ţ�ԭ�ӵ�����������Ϊ100����������ɫ��ѧ��Ҫ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com