
| A����Һ������Ũ�ȿ������㣺c(Na��)��c(OH��) ��c(H��)��c(CH3COO��) |
| B����Һ������Ũ��һ�����㣺c(Na��)�� c(CH3COO H)�� c(CH3COO��) |
| C������Һ�м����������ᣬ����Ũ�ȿ������㣺c(CH3COO��)��c(Na��)��c(H��)��c(OH��) |
| D������Һ������pH=11��NaOH��Һ��pH=3�Ĵ�����Һ�������϶��ɣ�����ʱ����ĵ��볣��Ϊ1.75��10��5�� |
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
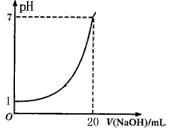
| A����Һ�����Ũ��Ϊ0��1 mol/L |
| B��NaOH��Һ��Ũ��Ϊ0��05 mol/L |
| C���ζ��յ�ʱ�����Ӽ�ʽ�ζ��ܶ���������NaOH��ҺŨ�Ȼ�ƫ�� |
| D��ָʾ����ɫʱ��˵��������NaOHǡ����ȫ��Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��C(s)��1/2O2(g)="CO(g)" ��H=-110.5kJ/mol |
| B��2H2(g)��O2(g)=2H2O(g)��H=-571.6kJ/mol |
| C��CH4(g)��2O2(g)=CO2(g)��2H2O(g) ��H=-890.3kJ/mol |
| D��C6H12O6(s)+6O2(g)=6CO2(g)+6H2O(l)��H=-2800kJ/mol |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��95 �洿ˮ��pH��7��˵�����ȿɵ���ˮ������ |
| B��pH = 3�Ĵ�����Һ��ϡ����10����pH = 4 |
| C��0.2 mol��L��1�����ᣬ������ˮ��Ϻ�pH = 1 |
| D��pH = 3�Ĵ�����Һ����pH = 11������������Һ�������Ϻ�pH = 7 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
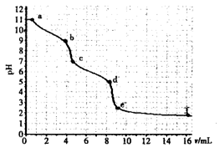
| A��̼���Ƶ�Ũ��Ϊ0.001mol��L��1 |
| B����pH=7ʱ��Һ��[Na��]=[Cl��] |
| C����pH=6ʱ��[Na+]��[HCO3��]��[CO32��] |
| D��c��d��������Ҫ���ӷ�Ӧ��CO32��+H+=HCO3�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A��ˮ���ӵı���ģ���ǣ� |
| B��100��ʱ����ˮ��pH=7 |
| C����Ȼˮ�к���2H2O����ˮ�� |
| D��1H2O��2H2O�������ʡ���ѧ������ȫ��ͬ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��c(Na��)��c(CH3COO��)��c(H+)��c(OH��) |
| B��c(Na��)��c(OH��)��c(CH3COO��)��c(H+) |
| C��c(Na��) + c(H+)=c(CH3COO��) + c(OH��) |
| D��c(Na��) + c(OH��)=c(CH3COO��) + c(H+) |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com