
(1)д������ȼ�յ��Ȼ�ѧ����ʽ____________________________��
(2)�� 1 mol CH4������ȫȼ������CO2�����ˮ�������ų�����Ϊ_____890 kJ(���������������)��
(3)����a mol CH4��CO��H2�Ļ��������ȫȼ�գ����� CO2�����Һ̬ˮʱ����ų�����(Q)��ȡֵ��Χ��____________________________��
(4)����a mol CH4��CO��H2�Ļ��������ȫȼ�գ����� CO2�����Һ̬ˮ����CO2��ˮ�����ʵ������ʱ����ų�����(Q)��ȡֵ��Χ��____________________________��
(1)H2(g)+1/2O2(g)![]() H2O(l)����H=-286 kJ��mol-1
H2O(l)����H=-286 kJ��mol-1
(2)��
(3)283a kJ��Q��890a kJ
(4)284.5a kJ��Q��586.5a kJ
������������ȼ�յ��Ȼ�ѧ����ʽ��Ϊ1 mol ������ȫȼ������Һ̬ˮ�ų�������286 kJ����1 mol CH4������ȫȼ������ˮ�����ų�������������Һ̬ˮʱ�ų��������٣���a mol CH4��CO��H2���������ȫȼ�յ��������ü�ֵ�����ɵ�283a kJ��Q��890a kJ����a mol CH4��CO��H2���������ȫȼ�գ���CO2��ˮ�����ʵ�����ȣ���ʱ���з��̼����284.5a kJ��Q��586.5a kJ��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��֪1 mol CO������ȫȼ������CO2����ų�283 kJ������1 mol������ȫȼ������Һ̬ˮ�ų�286 kJ������1 mol CH4������ȫȼ������CO2�����Һ̬ˮ�ų�890 kJ������
(1)д��������ȫȼ������Һ̬ˮ���Ȼ�ѧ��Ӧ����ʽ��____________________________��
1 mol CH4������ȫȼ������CO2�����ˮ�������ų�������_____________890 kJ(�������=������)��
(2)������ˮú���е������ɷ֣�����ͬ״����Ҫ�õ���ȵ�������������ˮú�������������ԼΪ_______������ȼ�����ɵ�CO2������֮��Ϊ______________(ǰ�߱Ⱥ���)��
(3)�������ݺͼ���˵��������Ȼ������ˮú��������ȼ�ϣ�ͻ�����ŵ���______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���������䡱�������������ص㹤�̣����������ָ��Ȼ������ҵ�Ͻ�̼��ˮ�ڸ����·�Ӧ�Ƶ�ˮú��������CO��H2�������Ϊ1��1��
��֪1 mol CO������ȫȼ������CO2����ų�283 kJ������1 mol������ȫȼ������Һ̬ˮ�ų�286 kJ������1 mol CH4������ȫȼ������CO2�����Һ̬ˮ�ų�890 kJ������
(1)д��������ȫȼ������Һ̬ˮ���Ȼ�ѧ��Ӧ����ʽ��
________________________________________________________________________________
1 mol CH4������ȫȼ������CO2�����ˮ�������ų�������__________890 kJ(���������=������)��
(2)������ˮú���е������ɷ֣�����ͬ״����Ҫ�õ���ȵ�������������ˮú�������������Ϊ__________������ȼ�����ɵ�CO2������֮��Ϊ__________(ǰ�߱Ⱥ���)��
(3)�������ݺͼ���˵��������Ȼ������ˮú��������ȼ�ϣ�ͻ�����ŵ���_______________
_______________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��9�֣����������䡱�������������ص㹤�̣����������ָ��Ȼ��������Ҫ�ɷ��Ǽ��顣��ҵ�Ͻ�̼��ˮ�ڸ����·�Ӧ�Ƶ�ˮú����ˮú������Ҫ�ɷ���CO��H2�����ߵ������ԼΪ1��1����֪1 mol CO������ȫȼ������CO2����ų�283 kJ������1 mol������ȫȼ������Һ̬ˮ�ų�286 kJ������1 mol CH4������ȫȼ������CO2�����Һ̬ˮ�ų�890 kJ������
��1��д��������ȫȼ������Һ̬ˮ���Ȼ�ѧ��Ӧ����ʽ��_____________________ ��
��1 mol CH4������ȫȼ������CO2�����ˮ�������ų�������_______890 kJ���>������=����<��)��
��2������ˮú���������ɷ֣���ͬ״�������õ���ȵ�����������ˮú�������������ԼΪ_______��ȼ�����ɵ�CO2��������ԼΪ_______��
��3���������ݺͼ���˵��������Ȼ������ˮú��������ȼ�ϣ�ͻ�����ŵ���___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012����ױ���������ʯ��ҵһ�и�һ��ѧ����ĩ���Ի�ѧ�Ծ����������� ���ͣ������
��6�֣���2CO(g)��O2(g) 2CO2(g)��Ӧ���̵������仯��ͼ��ʾ����֪1 mol CO (g)����Ϊ1 mol CO2 (g)�Ħ�H ����283 kJ/mol����ش��������⣺
2CO2(g)��Ӧ���̵������仯��ͼ��ʾ����֪1 mol CO (g)����Ϊ1 mol CO2 (g)�Ħ�H ����283 kJ/mol����ش��������⣺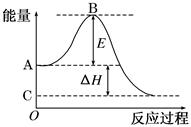
(1)ͼ��A���ʾ��____________________________________��E�Ĵ�С�Ը÷�Ӧ�ķ�Ӧ��________(��С����ޡ�)Ӱ�졣
(2)ͼ�Ц�H �� ________kJ/mol��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012����ױ����и�һ��ѧ����ĩ���Ի�ѧ�Ծ��������棩 ���ͣ������
��6�֣���2CO(g)��O2(g) 2CO2(g)��Ӧ���̵������仯��ͼ��ʾ����֪1 mol CO
(g)����Ϊ1 mol CO2
(g)�Ħ�H ����283 kJ/mol����ش��������⣺
2CO2(g)��Ӧ���̵������仯��ͼ��ʾ����֪1 mol CO
(g)����Ϊ1 mol CO2
(g)�Ħ�H ����283 kJ/mol����ش��������⣺

(1)ͼ��A���ʾ��____________________________________��E�Ĵ�С�Ը÷�Ӧ�ķ�Ӧ��________(��С����ޡ�)Ӱ�졣
(2)ͼ�Ц�H �� ________kJ/mol��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com