
ЁОЬтФПЁПAЃЌBЃЌCЃЌDЫФжжЖЬжмЦкдЊЫиЕФдзгАыОЖвРДЮМѕаЁЃЌDФмЗжБ№гыAЃЌBЃЌCаЮГЩЕчзгзмЪ§ЯрЕШЕФЗжзгXЁЂYЁЂZЁЃCдзгЕФзюЭтВуЕчзгХХВМЮЊnsnnp2nЁЃEЕФдзгађЪ§ЮЊ29ЁЃ
(1)AЃЌBЃЌCЕФЕквЛЕчРыФмгЩаЁЕНДѓЕФЫГађЮЊ________(гУдЊЫиЗћКХБэЪО)ЁЃ
(2)XЪЧКЌга________Мќ(ЬюЁАЗЧМЋадЁБЛђЁАМЋадЁБЃЌЯТЭЌ)ЕФ________ЗжзгЁЃ
(3)AЕФвЛжжЧтЛЏЮяЕФЯрЖдЗжзгжЪСПЮЊ26ЃЌЦфЗжзгжаЕФІвМќгыІаМќЕФМќЪ§жЎБШЮЊ________ЁЃ
(4)YЗжзгЕФПеМфЙЙаЭЮЊ__________ЃЌЦфжааФдзгВЩШЁ________дгЛЏЁЃ
(5)вЛжжгЩBЃЌCзщГЩЕФЛЏКЯЮягыAC2ЛЅЮЊЕШЕчзгЬхЃЌЦфЛЏбЇЪНЮЊ________ЁЃ
(6)YЪЧвЛжжвзвКЛЏЕФЦјЬхЃЌЧыМђЪіЦфвзвКЛЏЕФдвђ_________ЁЃ
(7)аДГіE2ЃЋЕФЕчзгХХВМЪН___________________ЃЌВЂаДГіE2ЃЋдкZжаЭЈШызуСПYЕУЕНЩюРЖЩЋШмвКЕФРызгЗДгІЗНГЬЪН_______ЁЃ
ЁОД№АИЁПC<O<N МЋад ЗЧМЋад 3:2 Ш§НЧзЖаЮ sp3 N2O АБЦјЗжзгМфДцдкЧтМќЃЌЗаЕуИпЖјШнвзвКЛЏ 1s22s22p63s23p63d9 Cu2++4NH3H2O=[Cu(NH3)4]2++4H2O
ЁОНтЮіЁП
AЁЂBЁЂCЁЂDЫФжжЖЬжмЦкдЊЫиЕФдзгАыОЖвРДЮМѕаЁЃЌDФмЗжБ№гыAЃЌBЃЌCаЮГЩЕчзгзмЪ§ЯрЕШЕФЗжзгXЁЂYЁЂZЃЌЫЕУїDЮЊЧтдЊЫиЃЌвђЮЊsВужЛФмЪЧ2ИіЕчзгЃЌЫљвдn=2ЃЌМДCдзгЕФзюЭтВуЕчзгХХВМЮЊ2s22p4ЃЌЮЊбѕдЊЫиЃЌдђAЮЊЬМЃЌBЮЊЕЊЃЌдђаЮГЩЕФЧтЛЏЮяЗжБ№ЮЊМзЭщЁЂАБЦјКЭЫЎЁЃEЕФдзгађЪ§ЮЊ29ЃЌЮЊЭдЊЫиЁЃ
(1)ЭЌжмЦкЫцзХдзгађЪ§діДѓдЊЫиЕФЕквЛЕчРыФмГЪдіДѓЧїЪЦЃЌЕЋЕЊдЊЫиЕФ2pФмМЖШнФЩ3ИіЕчзгЃЌЮЊАыТњЮШЖЈзДЬЌЃЌФмСПНЯЕЭЃЌЕквЛЕчРыФмгЩаЁЕНДѓЕФЫГађЮЊC<O<NЁЃ(2)XЪЧМзЭщЃЌКЌгаМЋадМќЃЌЮЊе§ЫФУцЬхНсЙЙЃЌЗжзгжае§ИКЕчКЩжиаФжиКЯЃЌЪєгкЗЧМЋадЗжзгЁЃ(3)AЕФвЛжжЧтЛЏЮяЕФЯрЖдЗжзгжЪСПЮЊ26ЃЌИУЗжзгЮЊввШВЃЌЦфНсЙЙЪНЮЊ![]() ЃЌЬМЧтЕЅМќЮЊІвМќЃЌЬМЬМШ§МќКЌгавЛИіІвМќКЭ2ИіІаМќЃЌЙЪЦфЗжзгжаЕФІвМќКЭІаМюЕФМќЪ§жЎБШЮЊ3:2ЁЃ(4)YЗжзгЮЊАБЦјЃЌПеМфЙЙаЭЮЊШ§НЧзЖаЮЃЌЦфжааФдзгЕЊдзгМлВуЕчзгЖдЪ§=3+ЃЈ5-1ЁС3ЃЉ/2=4ЃЌВЩШЁsp3дгЛЏЁЃ(5)вЛжжгЩЕЊКЭбѕзщГЩЕФЛЏКЯЮягыЖўбѕЛЏЬМЛЅЮЊЕШЕчзгЬхЃЌЦфЛЏбЇЪНЮЊN2OЁЃ(6)АБЦјЗжзгМфДцдкЧтМќЃЌЗаЕуИпЖјШнвзвКЛЏЁЃ (7)ЭРызгЕФЕчзгХХВМЪН1s22s22p63s23p63d9ЃЌЭРызгдкЫЎжаЭЈШызуСПАБЦјЕУЕНЩюРЖЩЋШмвКЕФРызгЗДгІЗНГЬЪНЮЊCu2++4NH3H2O=[Cu(NH3)4]2++4H2OЁЃ
ЃЌЬМЧтЕЅМќЮЊІвМќЃЌЬМЬМШ§МќКЌгавЛИіІвМќКЭ2ИіІаМќЃЌЙЪЦфЗжзгжаЕФІвМќКЭІаМюЕФМќЪ§жЎБШЮЊ3:2ЁЃ(4)YЗжзгЮЊАБЦјЃЌПеМфЙЙаЭЮЊШ§НЧзЖаЮЃЌЦфжааФдзгЕЊдзгМлВуЕчзгЖдЪ§=3+ЃЈ5-1ЁС3ЃЉ/2=4ЃЌВЩШЁsp3дгЛЏЁЃ(5)вЛжжгЩЕЊКЭбѕзщГЩЕФЛЏКЯЮягыЖўбѕЛЏЬМЛЅЮЊЕШЕчзгЬхЃЌЦфЛЏбЇЪНЮЊN2OЁЃ(6)АБЦјЗжзгМфДцдкЧтМќЃЌЗаЕуИпЖјШнвзвКЛЏЁЃ (7)ЭРызгЕФЕчзгХХВМЪН1s22s22p63s23p63d9ЃЌЭРызгдкЫЎжаЭЈШызуСПАБЦјЕУЕНЩюРЖЩЋШмвКЕФРызгЗДгІЗНГЬЪНЮЊCu2++4NH3H2O=[Cu(NH3)4]2++4H2OЁЃ



| ФъМЖ | ИпжаПЮГЬ | ФъМЖ | ГѕжаПЮГЬ |
| ИпвЛ | ИпвЛУтЗбПЮГЬЭЦМіЃЁ | ГѕвЛ | ГѕвЛУтЗбПЮГЬЭЦМіЃЁ |
| ИпЖў | ИпЖўУтЗбПЮГЬЭЦМіЃЁ | ГѕЖў | ГѕЖўУтЗбПЮГЬЭЦМіЃЁ |
| ИпШ§ | ИпШ§УтЗбПЮГЬЭЦМіЃЁ | ГѕШ§ | ГѕШ§УтЗбПЮГЬЭЦМіЃЁ |
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПдкВЛЭЌЧщПіЯТВтЕУA(g)ЃЋ3B(g)![]() 2C(g)ЃЋ2D(g)ЕФЯТСаЗДгІЫйТЪЃЌЦфжаЗДгІЫйТЪзюДѓЕФЪЧ
2C(g)ЃЋ2D(g)ЕФЯТСаЗДгІЫйТЪЃЌЦфжаЗДгІЫйТЪзюДѓЕФЪЧ
A. Ід(D)=0.01 molL-1s-1 B. Ід(C)=0.5 molL-1min-1
C. Ід(B)=0.6 molL-1min-1 D. Ід(A)=0.2molL-1min-1
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСаЮяжЪжаЃЌМШФмгыЧПЫсЗДгІЃЌгжФмИњЧПМюЗДгІЕФЪЧЃЈ ЃЉ
Ђй![]() ЂкAl ЂлAl2O3Ђм
ЂкAl ЂлAl2O3Ђм![]() Ђн
Ђн![]()
A.ЂйЂкЂлЂмЂнB.ЂкЂлЂмC.ЂйЂлЂмD.ЂйЂкЂлЂм
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПI.ШчЭМЫљЪОЮЊГЃМћВЃСЇвЧЦїЕФВПЗжНсЙЙЃК
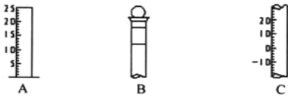
ЃЈ1ЃЉЧыаДГіЫљСавЧЦїЕФУћГЦЃКA__________ЃЌB____________;C_____________ЃЛ
ЃЈ2ЃЉвЧЦї B ЪЙгУЧАБиаы______________________ЁЃ
ЃЈ3ЃЉвЧЦї B ЩЯБъМЧга__________________(ЬюађКХ)ЃЛ
ЂйжЪСП ЂкЮТЖШ ЂлПЬЖШЯп ЂмХЈЖШ ЂнШнЛ§
IIЃЎ ФГЮоЩЋЙЄвЕЗЯЫЎжаПЩФмКЌгаNa+ЁЂMg2+ЁЂAl3+ЁЂCl-ЁЂSO42-жаЕФМИжжРызгЁЃ
aЃЎШЁЩйаэИУЗЯЫЎгкЪдЙмжаЃЌМгШызуСПЕФBa(NO3)2ШмвККЭЯЁЯѕЫсЃЌВњЩњАзЩЋГСЕэЃЌГфЗжЗДгІКѓЙ§ТЫЃЌЯђТЫвКжаМгШыAgNO3ШмвКЮоГСЕэВњЩњЁЃ
b.СэШЁ10mLИУЗЯЫЎгкЪдЙмжаЃЌЕЮМгNaOHШмвКЯШВњЩњАзЩЋГСЕэЃЌКѓВПЗжГСЕэШмНтЁЃЩњГЩГСЕэЕФЮяжЪЕФСПЫцМгШыNaOHЕФЮяжЪЕФСПЙиЯЕШчЯТЭМЫљЪОЁЃ
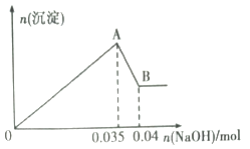
ИљОнЩЯЪіЪЕбщКЭЭМжаЪ§ОнЃК
ЃЈ1ЃЉИУЗЯЫЎжавЛЖЈВЛКЌгаЕФРызгга________________(ЬюРызгЗћКХ)ЃЛ
ЃЈ2ЃЉаДГіAЁњBЙ§ГЬжаЗЂЩњЗДгІЕФРызгЗНГЬЪНЃК_________________ЃЛ
ЃЈ3ЃЉИУЗЯЫЎжаЃЌc(Al3+)=________________________ЃЛ
ЃЈ4ЃЉЂйЪЕбщЪвХфжЦ100mL2mol/LЕФNaOHШмвКЪБЃЌгУЕНЕФВЃСЇвЧЦїГ§ЩеБЁЂВЃСЇАєЁЂСПЭВЭтЃЌЛЙашвЊ________(ЬювЧЦїУћГЦ)ЁЃ
ЂкЯТСаВйзїЪЙЫљХфШмвКХЈЖШЦЋДѓЕФЪЧ(ЬюаДзжФИ)________ЁЃ
AЃЎГЦСПгУСЫЩњатЕФэРТы
B.НЋNaOHЗХдкжНеХЩЯГЦСП
C.NaOHдкЩеБжаШмНтКѓЃЌЮДРфШДОЭСЂМДзЊвЦЕНШнСПЦПжа
D.ЭљШнСПЦПзЊвЦЪБЃЌгаЩйСПвКЬхМњГі
EЃЎЮДЯДЕгШмНтNaOHЕФЩеБ
FЃЎЖЈШнЪБбіЪгПЬЖШЯп
GЃЎШнСПЦПЮДИЩдяМДгУРДХфжЦШмвК
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСаЕчРыЗНГЬЪНЪщаДДэЮѓЕФЪЧ
A. MgCl2ЃНMg2++2Cl B. NaOHЃНNa++O2+H+
C. HClЃНH+ +Cl D. K2SO4ЃН2K++SO42
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПгаЮДжЊХЈЖШЕФBa(OH)2КЭNaOHЛьКЯШмвКЃЌСПШЁЫФЗнИУШмвКЗжБ№ЭЈШыЕШСПЕФCO2ЃЈвбелЫуГЩБъзМзДПіЯТЕФЬхЛ§ЃЌВЛПМТЧCO2дкЫЎжаЕФШмНтЃЉЃЌЩњГЩГСЕэЕФЮяжЪЕФСПШчЯТБэЃК
ЪЕбщађКХ | Ђё | Ђђ | Ђѓ | Ђє |
CO2ЬхЛ§(mL) | 2352 | 2352 | 2352 | 2352 |
бљЦЗЬхЛ§(mL) | 20.0 | 30.0 | 40.0 | 50.0 |
ГСЕэЮяжЪЕФСП(ЁС10-2mol) | 1.50 | 4.50 | 6.00 | 7.50 |
ЃЈ1ЃЉИУЛьКЯШмвКжаBa(OH)2ЕФЮяжЪЕФСПХЈЖШ=_____________ЁЃ
ЃЈ2ЃЉЪЕбщЂѓзюКѓЫљЕУШмвКжаЬМЫсФЦЕФЮяжЪЕФСП=___________ЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПГЃЮТЯТЃЌНЋCl2ЛКТ§ЭЈШыЫЎжажСБЅКЭЃЌШЛКѓЯђЫљЕУБЅКЭТШЫЎжаЕЮМг0.10 molЁЄLЃ1ЕФNaOHШмвКЃЌећИіЙ§ГЬжаШмвКpHБфЛЏЕФЧњЯпШчЭМЫљЪОЁЃЯТСаа№Ъіжае§ШЗЕФЪЧ

A. ЕуЂйЫљЪОШмвКжаЃКc(H+)=c(ClЃ)+c(HClO)+c(OHЃ)
B. ЕуЂкЫљЪОШмвКжаЃКc(H+)ЃОc(ClЃ)ЃОc(ClOЁЊ)ЃОc(HClO)
C. ЕуЂлЫљЪОШмвКжаЃКc(Na+)=2c(ClOЃ)+c(HClO)
D. ЕуЂмЫљЪОШмвКжаЃКc(Na+)>c(ClOЃ)>c(ClЃ)>c(HClO)
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЩшNAЮЊАЂЗќМгЕТТоГЃЪ§ЕФжЕЃЌЯТСаЫЕЗЈе§ШЗЕФЪЧ
A. 56 g Fe гы1mol Cl2ГфЗжЗДгІЃЌзЊвЦЕФЕчзгЪ§ЮЊ3NA
B. ЗжзгЪ§ЮЊ 0.1NAЕФCH4КЭNH3ЛьКЯЦјЬхЃЌКЌгаЕФЙВгУЕчзгЖдЪ§ЮЊ0.4NA
C. 25ЁцЪБЃЌ1L pH=12ЕФBa(OH)2ШмвКжаКЌга0.02NA ЕФOHЃ
D. ГЃЮТГЃбЙЯТЃЌ22 g D3O+жаКЌга10NAИіЕчзг
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСаЮяжЪзЊЛЏдкИјЖЈЬѕМўЯТУПвЛВНЖМФмЪЕЯжЕФЪЧ
A. Si![]() SiO2
SiO2![]() H2SiO3
H2SiO3
B. Mg(OH)2![]() MgCl2(aq)
MgCl2(aq) ![]() Mg
Mg
C. Al2O3![]() A1Cl3(aq)
A1Cl3(aq) ![]() ЮоЫЎAlCl3
ЮоЫЎAlCl3
D. CH3CHO![]() CH3COOH
CH3COOH![]() CH3COOCH3
CH3COOCH3
ВщПДД№АИКЭНтЮі>>
АйЖШжТаХ - СЗЯАВсСаБэ - ЪдЬтСаБэ
КўББЪЁЛЅСЊЭјЮЅЗЈКЭВЛСМаХЯЂОйБЈЦНЬЈ | ЭјЩЯгаКІаХЯЂОйБЈзЈЧј | ЕчаХеЉЦОйБЈзЈЧј | ЩцРњЪЗащЮожївхгаКІаХЯЂОйБЈзЈЧј | ЩцЦѓЧжШЈОйБЈзЈЧј
ЮЅЗЈКЭВЛСМаХЯЂОйБЈЕчЛАЃК027-86699610 ОйБЈгЪЯфЃК58377363@163.com