
ij�о�С����CaCl2��H2Ϊԭ�ϣ���ͼ�Ʊ� +1��Ca�Ļ����������ֲ�����ֻ�����ֻ�������ң���Ԫ����ɷ���������������иơ���Ԫ�ص����������ֱ�Ϊ52.36%��46.33%���������ҵ�ˮ��Һ�����ԡ���ش��������⣺
��1�����о�С���Ƿ�ɹ��Ƶ� +1��Ca�Ļ���� ����ǡ������Ļ�ѧʽ�� ��
��2������ˮ��Ӧ�ɵ�H2���仯ѧ����ʽ�� ����Ӧ������Һ���ᾧ�ɵõ�һ�־��壬�仯ѧʽΪCaCl2�� xCa(OH)2�� 12H2O��Ϊȷ��x��ֵ�������ʵ�鷽�� ��
��3���ڼ��������£��ҵ�ˮ��Һ��Ũ����MnO2��Ӧ�����ӷ���ʽ�� ���ҵ�ˮ��Һ��Fe��Ӧ���õ���Һ���ȶ����������Һ�Ĵ�ʩ�� ��
��4����д��һ������Ϊ���ܵõ�CaCl�Ļ�ѧ����ʽ����CaCl2Ϊԭ�ϣ� ��
��1���� ��1�֣�CaHCl ��2�֣�
��2��2CaHCl + 2H2O = CaCl2 + Ca(OH)2 + 2H2����2�֣�
ȡ������ϡHNO3�ܽ��ֳɶ��ȷݣ�����һ�ݼ���Na2CO3��Һ���õ�CaCO3���������غ����n(Ca2+)����һ�ݼ���AgNO3��Һ���õ�AgCl���������غ����n(Cl-)�� ����ʽ�������xֵ�� ��3�֣�����������Ҳ���֣�
��3�֣�����������Ҳ���֣�
��3��2Cl- + MnO2 + 4H+�� Mn2+ + Cl2��+ 2H2O ��2�֣�
����FeCl2��Һ�����ԣ����������۷�ֹ���� ��2�֣�
��4��Ca + CaCl2 = 2CaCl
���������������1����������иơ���Ԫ�ص����������ֱ�Ϊ52.36%��46.33%����������100%С�������������л�������Ԫ�أ�����������Ϊ��100%��52.36%��46.33%��1.31%����ˣ�������ȷ�����������ʵĻ�ѧʽ��
N(Ca)�UN(Cl)�UN(H)��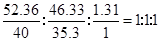
���Եó���ѧʽΪ��CaHCl�����Կ���û�еõ���ν����һ�۵ĸơ�
��2������ˮ��Ӧ�ɵ�H2��˵���Ƿ�������Ԫ�ص�����������ԭ��Ӧ��
2CaHCl + 2H2O = CaCl2 + Ca(OH)2 + 2H2��
ȡ������ϡHNO3�ܽ��ֳɶ��ȷݣ�����һ�ݼ���Na2CO3��Һ���õ�CaCO3���������غ����n(Ca2+)����һ�ݼ���AgNO3��Һ���õ�AgCl���������غ����n(Cl-)�� ����ʽ�������xֵ��
��3���ӵ�һ���п���֪����CaCl2��H2��CaHCl��HCl
2Cl- + MnO2 + 4H+��Mn2+ + Cl2��+ 2H2O
�ҵ�ˮ��Һ��Fe��Ӧ���õ���FeCl2��Һ�����ȶ���һ�����ױ������е��������������Ƕ��������ӻᷢ��ˮ�⣬�������Һ�Ĵ�ʩ�DZ���FeCl2��Һ�����ԣ����������۷�ֹ����
��4��������ǿ�Ļ�ԭ������ԭ����Ca + CaCl2 = 2CaCl
���㣺�������ʵIJⶨ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������������ȷ��Ԫ��X�����ԭ��������
��1�� 1.01��105 Pa,273 ��ʱ��̬����Xn���ܶ�Ϊd g/L����X�����ԭ������Ϊ_____��
��2����ͬ״���£�һ�������X����̬�⻯��HmX�������ǵ����NH3��2������X�����ԭ������Ϊ_____��
��3��a��Xԭ�ӵ�������Ϊb g����X�����ԭ�������ɱ�ʾΪ_____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
CO�dz����Ļ�ѧ���ʣ��й������ʺ�Ӧ�õ��о����¡�
��1����ͬѧ��ΪCO���Ա����Ը��������Һ����ΪCO2���������ʵ����֤��ͬѧ�IJ����Ƿ���ȷ��������������ͼ��������������ҩƷ�����ƻ�ѧʽ���Ӷ����������ƣ��������Բ���Ҳ���Բ�������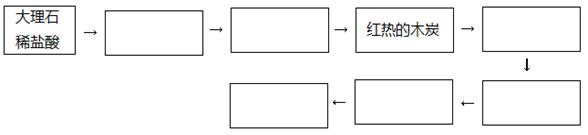
��2��CO��������һ�������¿��Ժϳ����ʻ�����[Fe(CO)5]�������ʿ�������Ǧ���͵ķ���������һ��dz��ɫҺ�壬�۵㡪20.5�棬�е�103�棬�����ڱ����л��ܼ���������ˮ���ܶ�1.46��1.52g/cm3���ж�������ʱ����Fe2(CO)9��60�淢����ȼ�����ʻ��������Ʊ�ԭ�����£�
Fe(s)+5CO(g) Fe(CO)5(g)
Fe(CO)5(g)
������˵����ȷ���� ��
| A������������Ӧԭ�����Ʊ��ߴ��� |
| B���Ʊ�Fe(CO)5Ӧ�ڸ��������������½��� |
C����ӦFe(s)+5CO(g) Fe(CO)5(g)��ƽ�ⳣ������ʽΪ Fe(CO)5(g)��ƽ�ⳣ������ʽΪ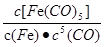 |
| D��Fe(CO)5Ӧ�ܷ⡢�������ܹⲢ����������ˮҺ������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������Ҫ�ɷ���FeS2��ij���᳧�ڽ��л�����ɷֲⶨʱ��ȡ0.1000 g��Ʒ�ڿ����г�����գ������ɵ�SO2����������Fe2(SO4)3��Һ��ȫ��Ӧ����Ũ��Ϊ0.02000 mol��L��K2Cr2O7����Һ�ζ����յ㣬����K2Cr2O7��Һ25.00 mL ��
��֪��SO2 + 2Fe3+ + 2H2O��SO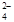 + 2Fe2+ + 4H+
+ 2Fe2+ + 4H+
Cr2O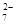 + 6 Fe2+ + 14 H+ ��2 Cr3+ + 6 Fe3+ + 7 H2O
+ 6 Fe2+ + 14 H+ ��2 Cr3+ + 6 Fe3+ + 7 H2O
��1����Ʒ��FeS2�����������ǣ��������ʲ��μӷ�Ӧ�� ��
��2��������6 g FeS2������SO2ȫ��ת��ΪSO3����ʱ�ų�9.83 kJ������������SO3��ˮȫ����������H2SO4���ų�13.03kJ������д��SO3����ת��ΪH2SO4���Ȼ�ѧ����ʽ�� ��
��3������10 t���������������ϲ���SO2���������״����Ϊ ���Ƶ�98������������Ϊ t ��SO2ȫ��ת��ΪH2SO4ʱ�ų��������� kJ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(1)��ͬ���ʵ�����O2��O3������������Ŀ֮��Ϊ_______,������ԭ�ӵ����ʵ���֮��Ϊ_________��
(2)�ڱ�״���£���4 g H2����11.2 L O2����1 mol H2O�У���ԭ����������_ ___�������С����_______��������ţ�
(3)����£���224L��HCl��������835ml����=1g/cm3����ˮ�У�����������ܶ�Ϊ1.2g/cm3�� ����������ʵ���Ũ��___________.��
(4)ij������������Ϊ6.4 g������6.02��1022�����ӣ�����������Է�������Ϊ_______��
(5)Na2CO3��Ħ��������____________��0.5mol Na2CO3��������____________������________mol Na+��Na+�ĸ���ԼΪ___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
0.5mol H2SO4��������________g����______________�����ӣ���________mol��ԭ�ӣ��ܺ�________mol NaOH��ȫ��Ӧ��������������Ԫ�ص�������________g��������Ԫ�ص�������ͬ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij���������ʵ���о�ʱ��������Ũ�Ⱦ�Ϊ1.0mol?L-1 ��Ba(OH)2��H2SO4��Һ��
��.����ͬѧ������Ba(OH)2��Һʱ��ֻ�ҵ��ڿ����б�¶�Ѿõ�Ba(OH)2��8H2O�Լ�����Է���������315����������ȡ�Լ���ˮ�н������ܽ⣬�ձ��д��ڴ���δ���
��1���û�ѧ����ʽ����δ���������ԭ��
��2��ijͬѧ���Ba(OH)2��8H2O��283K��293K��303Kʱ���ܽ�ȣ�g/100g H2O���ֱ�Ϊ2.5��3.9��5.6���ݴ���Ϊ����ʹ�ô�����Ba(OH)2��8H2O����������Ҳ�������1.0 mol?L-1Ba(OH)2��Һ���������ǣ�
��.��18 mol?L-1��Ũ��������450 mL 1.0 mol?L-1ϡ���ᡣ
(3)ʵ��ʱ������ȡŨ��������Ϊ mL�����õ���Ҫ��������Ͳ���ձ����������� ��
(4)������Һʱ�����ݵIJ��������ǣ�
(5)ʵ�������������ʹ������ҺŨ��ƫ�ߵ���
| A��ϴ����ȡŨ�������Ͳ2��3�Σ�����ϴ��Һת������ƿ�� |
| B������ʱ���ӿ̶��� |
| C����ҡ�Ⱥ������¼�ˮ���̶��� |
| D����ˮϴ������ƿδ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��״���£�1���ˮ�����ܽ�500�����HCl���塣����1 Lˮ��ͨ���״���µ�448LHCl���壬����������ȫ�ܽ⡣
��1����������Һ�ܶ�Ϊ1.2 g/cm3������Һ�к�HCl���ʵ���Ũ��Ϊ ��
��2���Ӹ���Һ��ȡ��10mLŨ�����ܽ���ˮ���Ƴ�500mL��Һ�����ƺ��ϡ��Һ�к�HCl���ʵ���Ũ��Ϊ ��
��3������Ũ������������ϡ����ʱ�����������У�ʹ��ǰ�������Ƿ�©Һ�������� �����ƹ����У����Ũ��ƫ�͵IJ���������_______________��ѡ�����в�������ţ���
| A������ƿ����ˮϴ��δ�Ӹ��� |
| B����Ͳ������ˮϴ��δ���� |
| C�����ձ���Ũ������������ƿ��δ��ˮϴ���ձ������������м�ˮ���̶� |
| D���ý�ͷ�ι�������ƿ�м�ˮʱ�����������̶��ߣ������⽺ͷ�ιܴ�ƿ������������Һʹʣ����Һ���ɴ�̶��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
�����仯���������������е�Ӧ�ù㷺��
��1��Al4C3����ұ�����������ᷴӦ������֮һ�Ǻ�������ߵ������Ļ�ѧ����ʽ�ǣ� ��14.4g Al4C3�����ᷴӦ������������ L����״������
��2��AlN���ڵ���������AlN�г���������̼����һ����������̼��AlN��Ʒ�����ܱշ�Ӧ���У�ͨ��4.032L����״���µģ�O2���ڸ����³�ַ�Ӧ����������ܶ�Ϊ1.34g/L��������ɱ�״����AlN����O2��Ӧ���������������Ħ������Ϊ g/mol��������ijɷ��� ��д��ѧʽ��������Ʒ�к�����̼ g��
��3�������Ӳ��þ��ˮ��ʯ��һ�����͵����ӽ������ϡ��Ʊ�����ˮ��ʯ�Ĺ����ǣ�MgCl2��AlCl3��NaOH��NaCl��Һ����һ��������ϣ���65���ַ�Ӧ�����ˡ�ϴ�ӡ�����õ���ˮ��ʯ��Ϊȷ����ˮ��ʯ�ijɷ֣��������²�����
��ȡ26.65g��Ʒ���ڸ�����ʹ���ַֽ⣬�õ���������������壬��������ͨ��������Ũ�����Ũ����������Һ��������Һ��ֱ�����9.9g��3.65g������������������ɫ���������գ���������ɫ��
����ȡ26.65g��Ʒ������������ϡ���ᣬʹ����ȫ�ܽ⣬�ټ���NaOH��Һ�����������յõ�11.6g��ɫ������ͨ������ȷ�������Ӳ��þ��ˮ��ʯ�Ļ�ѧʽ��
��4�������Ӳ��þ��ˮ��ʯ�ڿ����з��ã�������CO2��Ӧ�����������ӻᱻ̼������Ӵ��档26.65g��Ʒ�ڿ����з���һ��ʱ���������Ϊ25.925g����̼������Ӵ��沿�������ӵ�ˮ��ʯ�Ļ�ѧʽ�� ���˹��̵Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com