
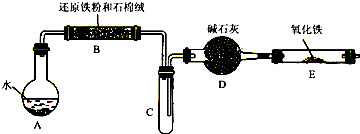
ѧ��������ͼ����װ�ý��С�����ˮ������Ӧ����ʵ�飬�����ò����һ����ȡFeCl3��6H2O���塣��ͼ�мгּ�β������װ�þ�����ȥ��
(1) �ش��������⣺�����������������ʣ�����ȥ���������л��е����ۿ���ѡ�õ��Լ�Ϊ__ __������ţ���
A��ϡ���� B������������Һ C��Ũ���� D. FeCl3��Һ
�˲��漰��Ӧ�����ӷ���ʽΪ �������Ӻ�����۽�һ�������װ�뷴Ӧ������
��2����Ӧ������װ��B�з�����Ӧ�Ļ�ѧ����ʽ��_______________ ____��
Dװ�õ����ã�________________.
��3����С��ѧ����B�з�Ӧ��IJ���������������ᣬ���ˣ���������Һ��ȡFeCl3��6H2O���壬����������£�
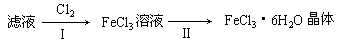
�������ӷ���ʽ��ʾ����I��ͨ��Cl2������ (2��)
���õ����ű�ʾ����ת�Ƶķ������Ŀ��
��Ϊ�˼���ijδ֪��Һ�Ƿ���FeCl2��Һ��ͬѧ�����������ʵ�鷽������֤����
��һ֧װ�и�δ֪��Һ���Թ�����ͨ���������ٵμ�KSCN��Һ����Һ���ֺ�ɫ��֤����δ֪��Һ��FeCl2��Һ������Ϊ�˷����Ƿ���� (���������������)��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
| Cl2 |
| I |
| II |


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
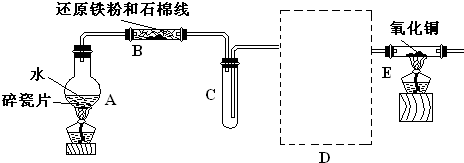
��15�֣�ijУ��ѧС��ѧ��������ͼ����װ�ý��� ������ˮ��Ӧ����ʵ�飬�����ò����һ����ȡFeCl3��6H2O���塣��ͼ�мгּ�β������װ�þ�����ȥ��
��1��װ��B�з�����Ӧ�Ļ�ѧ����ʽ��
��2��װ��E�е������ǣ�
��3��ֹͣ��Ӧ����B����ȴ��ȡ���еĹ��壬�������ϡ�����ַ�Ӧ�����ˡ�����������Һ�� Fe3+�IJ���������
��4����С��ѧ������������Һ��ȡFeCl3��6H2O���壬����������£�
�ٲ���I��ͨ��Cl2��������
�ڸ��������豣�������������Ҫԭ���ǣ�������ӷ���ʽ��Ҫ˵����
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com