
ʯīϩ�ɿ�����ʯī�IJ�״�ṹһ��һ��ذ����õ��ĵ���̼ԭ�ӣ�ʯīȲ��ƽ����״�ṹ��ȫ̼���ӣ����������Ļ�ѧ�ȶ��ԺͰ뵼�����ܣ��������⻯��ʯīϩ���е����Ǿ����У�ʹÿ��̼ԭ�Ӷ�����һ����ԭ�ӿɵ���ľ�Ե�²���ʯī�顣�����й�˵������ȷ����
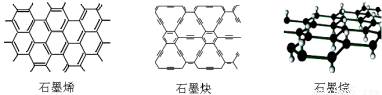
A��ʯīϩ��ʯīȲ��ʯī�黥Ϊͬ��������
B��12gʯīϩ��ȫת��Ϊʯī��������11.2L
C��ʯīȲ�����뵼����ϣ����������������
D��ʯīȲ���Դ���H2���ӵ�ֱ����������������H2�ᴿ��Ĥ
 ��ʦ����ɳ���ʱͬ��ѧ����ϵ�д�
��ʦ����ɳ���ʱͬ��ѧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ��������ݸ۾��ÿ�������ѧ��һ��12���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
�����Ũ�����У����н��������ܽ����
A���� B��þ C���� D��ͭ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016��㶫ʡ˳���и����ϵ��Ĵ��¿����ۻ�ѧ�Ծ��������棩 ���ͣ�ѡ����
�������ӷ���ʽ��ȷ����
A����ʯī���缫���CuCl2��Һ��2Cl-��2H2O��2OH-��Cl2��ʮH2��
B��˫��ˮ����ϡ�����KI��Һ��H2O2��2H����2I��=I2��2H2O
C��Na2O2����ˮ����O2��Na2O2��H2O=2Na����2OH����O2��
D��Na2S2O3��Һ�м���ϡ���2S2O32����4H��=SO42����3S����2H2O
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016�츣��ʡ������12���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
����ʵ�鷽������У��ܴﵽʵ��Ŀ�ĵ��ǣ�
ѡ�� | ʵ��Ŀ�� | ʵ�鷽�� |
A | ��֤��������Ư���� | ��������ͨ����ˮ�У�����Һ��ɫ�Ƿ���ɫ |
B | �����Ȼ�����Һ���Ƿ�FeCl2 | ���Ȼ�����Һ�еμ�����������Һ |
C | ����ʳ�����Ƿ�����KIO3 | ȡʳ������������KI��Һ�����������Һ���۲���Һ�Ƿ���� |
D | ��֤Ksp(AgI)��Ksp(AgCl) | �����ʵ���NaCl��KI�Ļ��Һ�еμ�AgNO3��Һ���۲������ɫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016������������������ѧ������ѧ�ڵ��Ĵ��¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
��NAΪ����٤��������ֵ������˵����ȷ����
A.��״���£�0.1mol Cl2����ˮ��ת�Ƶĵ�����ĿΪ0.1NA
B.���³�ѹ�£�18g H2O���е�ԭ������Ϊ3NA
C.��״���£�11.2L CH3CH2OH�к��з��ӵ���ĿΪ0.5NA
D.���³�ѹ�£�2.24L CO��CO2��������к��е�̼ԭ����ĿΪ0.1NA
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�꽭��ʡ��һ�ϵڶ����¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
Ҫ�Ӻ�Al3+��Fe3+��Ba2+��Ag+����Һ�зֱ������Fe3+��Ba2+��Ag+�������Լ���˳����ȷ����
A�����ᡢϡ���ᡢNaOH��Һ B��NaOH��Һ���ᡢϡ����
C�����ᡢϡ���ᡢ��ˮ D�����ᡢ��ˮ��ϡ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016�����ʡ������ѧ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
�����л���ֻ������ͬ���칹�����
A������ʽΪC4H10�����Ķ���ȡ����
B������ʽΪC3H9N���л������Nԭ������������������ԭ������
C���ұ������ϵĶ���ȡ����
D������ʽΪC4H8���л���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�����ʡ�߶��ϵ������¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
�����йص������Һ������Ũ�ȹ�ϵ��ȷ����
A��pH��1��NaHSO4��Һ��c(H��)��c(SO42��)��c(OH��)
B������AgCl��AgI���������Һ��c(Ag��)>c(Cl��)��c(I��)
C��CO2��ˮ��Һ��c(H��)>c(HCO3-)��2c(CO32-)
D���������ʵ�����NaHC2O4��Na2C2O4����Һ��3c(Na��)��2[c(HC2O4-)��c(C2O42-)��c(H2C2O4)]
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�꽭��ʡ��һ��ѧ�����п��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
�����Ƕ�ijˮ��Һ�������Ӽ���ķ����ͽ��ۣ�������ȷ����
A���ȼ���BaCl2��Һ���ټ���������HNO3��Һ�������˰�ɫ����������Һ��һ�����д�����SO42-
B������������CaCl2��Һ�������˰�ɫ����������Һ��һ�����д�����CO32-
C����������Ũ NaOH��Һ�������˴���ǿ�Ҵ̼�����ζ�����壬���������ʹʪ��ĺ�ɫʯ����ֽ����������Һ��һ�����д�����NH4+
D���ȼ����������Ὣ��Һ�ữ,�ټ�AgNO3��Һ�������˰�ɫ����������Һ��һ�����д�����Cl��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com