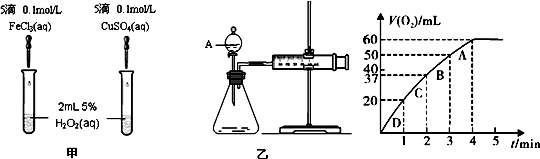
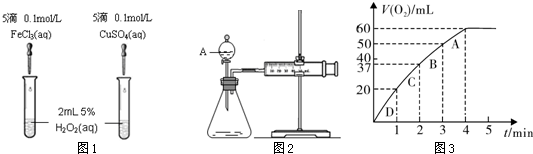
ͨ����ʵ��Ĺ۲졢�����������ó���ȷ�Ľ����ǻ�ѧѧϰ����Ҫ����֮һ��������ʵ����ʵ������Ľ�����ȷ���ǣ� ��?
A��KI������Һ��ͨ��Cl2����Һ����ԭ���ǵ�������Cl2������ɫ��Ӧ?
B��Ũ�����ڹ��������±��������Ũ����ȶ���������ɫ����������Ũ����?
C��ij��Һ�м��������ữ���Ȼ�����Һ���а�ɫ��������˵������Һ�к���SO42-
D�������£�Ũ����ɴ��������ʻ�����������˵����������������Ũ�����Ӧ?
���𰸡�������A��KI������Һ��ͨ��Cl2������I2����������I2������ɫ��Ӧ��
B��Ũ�����ڹ��������·ֽ�����NO2��
C��SO32-�ɱ���������ΪSO42-��
D����������Ũ���ᷢ���ۻ���Ӧ��
����⣺A��KI������Һ��ͨ��Cl2������I2��������I2����ɫ�������͵��۲���Ӧ����A����
B��Ũ�����ڹ��������·ֽ�����NO2��NO2����ˮ��������Һ��ƣ���B��ȷ��
C��SO32-�ɱ���������ΪSO42-�������ж�һ������SO42-����C����
D����������Ũ���ᷢ���ۻ���Ӧ����D����
��ѡB��
���������⿼�����ʵ����ʼ����飬��Ŀ�Ѷ��еȣ�����ע��������ʵ����ʵ�ѧϰ���������ʵļ�����