
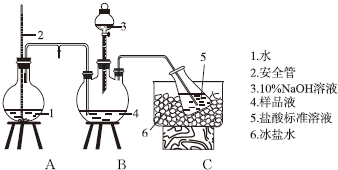
·ÖĪö £Ø1£©Ķعż2ÖŠŅŗĆęµ÷½ŚAÖŠŃ¹Ē棻
£Ø2£©¼īÖ»ÄÜŹ¢·ÅŌŚ¼īŹ½µĪ¶Ø¹ÜÖŠ£¬ĖįŠŌČÜŅŗÖ»ÄÜŹ¢·ÅŌŚĖįŹ½µĪ¶Ø¹ÜÖŠ£»NaOHČÜŅŗŗĶŃĪĖįČÜŅŗĒ”ŗĆ·“Ó¦ŗó³ŹÖŠŠŌ£¬æÉŅŌŃ”ŌńĖįŠŌ»ņ¼īŠŌ±äÉ«·¶Ī§ÄŚµÄÖøŹ¾¼Į£»
£Ø3£©øł¾Ż°±ĘųÓėŗĶŃĪĖį·“Ó¦Ö®¼äµÄ¹ŲĻµŹ½¼ĘĖć°±ĘųµÄÖŹĮ棬ŌŁøł¾ŻÖŹĮæ·ÖŹż¹«Ź½¼ĘĖć°±ÖŹĮæ·ÖŹż£»
£Ø4£©ČōĘųĆÜŠŌ²»ŗĆ£¬µ¼ÖĀ°±ĘųµÄĮæĘ«µĶ£»
£Ø5£©ĻõĖįŅų²»ĪČ¶Ø£¬¼ū¹āŅ×·Ö½ā£»øł¾ŻĄė×Ó»ż³£Źż¼ĘĖćc£ØCrO42-£©£»
£Ø6£©¾²ā¶Ø£¬ŃłĘ·XÖŠīÜ”¢°±ŗĶĀȵÄĪļÖŹµÄĮæÖ®±ČĪŖ1£ŗ6£ŗ3£¬ŌņĘä»ÆѧŹ½ĪŖ[Co£ØNH3£©6]Cl3£¬øł¾Ż»ÆŗĻĪļÖŠø÷ŌŖĖŲ»ÆŗĻ¼ŪµÄ“śŹżŗĶĪŖ0¼ĘĖćCoŌŖĖŲ»ÆŗĻ¼Ū£»øĆ·“Ó¦ÖŠCoŹ§µē×Ó”¢Ė«ŃõĖ®µĆµē×Ó£¬CoCl2•6H2O”¢NH4Cl”¢H2O2”¢NH3·¢Éś·“Ӧɜ³É[Co£ØNH3£©6]Cl3ŗĶĖ®£»Ė«ŃõĖ®Ņ×·Ö½ā”¢ĘųĢåµÄČܽā¶ČĖę×ÅĪĀ¶ČµÄÉżø߶ų½µµĶ£®
½ā“š ½ā£ŗ£Ø1£©ĪŽĀŪČż¾±ĘæÖŠŃ¹Ēæ¹ż“ó»ņ¹żŠ”£¬¶¼²»»įŌģ³ÉĪ£ĻÕ£¬Čō¹ż“ó£¬AŌŚµ¼¹ÜÄŚŅŗĆęÉżøߣ¬½«»ŗ³åŃ¹Į¦£¬Čō¹żŠ”£¬Ķā½ēæÕĘųĶعżµ¼¹Ü½ųČėÉÕĘ棬Ņ²²»»įŌģ³Éµ¹Īü£¬°²Č«×÷ÓƵÄŌĄķŹĒŹ¹AÖŠŃ¹ĒæĪČ¶Ø£¬
¹Ź“š°øĪŖ£ŗŹ¹AĘæÖŠŃ¹ĒæĪČ¶Ø£»
£Ø2£©¼īÖ»ÄÜŹ¢·ÅŌŚ¼īŹ½µĪ¶Ø¹ÜÖŠ£¬ĖįŠŌČÜŅŗÖ»ÄÜŹ¢·ÅŌŚĖįŹ½µĪ¶Ø¹ÜÖŠ£¬ĖłŅŌÓĆNaOH±ź×¼ČÜŅŗČ·¶Ø¹żŹ£µÄHClŹ±£¬Ó¦Ź¹ÓĆ¼īŹ½µĪ¶Ø¹ÜŹ¢·ÅNaOHČÜŅŗ£»NaOHČÜŅŗŗĶŃĪĖįČÜŅŗĒ”ŗĆ·“Ó¦ŗó³ŹÖŠŠŌ£¬æÉŅŌŃ”ŌńĖįŠŌ»ņ¼īŠŌ±äÉ«·¶Ī§ÄŚµÄÖøŹ¾¼Į£¬¼×»ł³ČĪŖĖįŠŌ±äÉ«ÖøŹ¾¼Į”¢·ÓĢŖĪŖ¼īŠŌ±äÉ«ÖøŹ¾¼Į£¬ĖłŅŌæÉŅŌєȔ¼×»ł³Č»ņ·ÓĢŖ×÷ÖøŹ¾¼Į£¬
¹Ź“š°øĪŖ£ŗ¼ī£»·ÓĢŖ»ņ¼×»ł³Č£»
£Ø3£©Óė°±Ęų·“Ó¦µÄn£ØHCl£©=V1”Į10-3L”ĮC1mol•L-1-C2mol•L-1 ”ĮV2”Į10-3L=£ØC1V1-C2V2£©”Į10-3mol£¬øł¾Ż°±ĘųŗĶHClµÄ¹ŲĻµŹ½ÖŖ£¬n£ØNH3£©=n£ØHCl£©=£ØC1V1-C2V2£©”Į10-3mol£¬°±µÄÖŹĮæ·ÖŹż=$\frac{£Ø{C}_{1}{V}_{1}-{C}_{2}{V}_{2}£©”Į1{0}^{-3}mol”Į17g/mol}{wg}$”Į100%£¬
¹Ź“š°øĪŖ£ŗ$\frac{£Ø{C}_{1}{V}_{1}-{C}_{2}{V}_{2}£©”Į1{0}^{-3}mol”Į17g/mol}{wg}$”Į100%£»
£Ø4£©ČōĘųĆÜŠŌ²»ŗĆ£¬µ¼ÖĀ²æ·Ö°±ĘųŠ¹Ā©£¬ĖłŅŌ°±ĘųÖŹĮæ·ÖŹżĘ«µĶ£¬¹Ź“š°øĪŖ£ŗĘ«µĶ£»
£Ø5£©ĻõĖįŅų²»ĪČ¶Ø£¬¼ū¹āŅ×·Ö½ā£¬ĪŖ·ĄÖ¹ĻõĖįŅų·Ö½ā£¬ÓĆ×ŲÉ«µĪ¶Ø¹ÜŹ¢·ÅĻõĖįŅųČÜŅŗ£»c£ØCrO42-£©=$\frac{K{\;}_{sp}}{{c}^{2}£ØA{g}^{+}£©}$=$\frac{1.12”Į1{0}^{-12}}{£Ø2.0”Į1{0}^{-5}£©^{2}}$mol/L=2.8”Į10-3 mol/L£¬
¹Ź“š°øĪŖ£ŗ·ĄÖ¹ĻõĖįŅų¼ū¹ā·Ö½ā£»2.8”Į10-3£»
£Ø6£©¾²ā¶Ø£¬ŃłĘ·XÖŠīÜ”¢°±ŗĶĀȵÄĪļÖŹµÄĮæÖ®±ČĪŖ1£ŗ6£ŗ3£¬ŌņĘä»ÆѧŹ½ĪŖ[Co£ØNH3£©6]Cl3£¬øł¾Ż»ÆŗĻĪļÖŠø÷ŌŖĖŲ»ÆŗĻ¼ŪµÄ“śŹżŗĶĪŖ0µĆCoŌŖĖŲ»ÆŗĻ¼ŪĪŖ+3¼Ū£»øĆ·“Ó¦ÖŠCoŹ§µē×Ó”¢Ė«ŃõĖ®µĆµē×Ó£¬CoCl2•6H2O”¢NH4Cl”¢H2O2”¢NH3·¢Éś·“Ӧɜ³É[Co£ØNH3£©6]Cl3ŗĶĖ®£¬·“Ó¦·½³ĢŹ½ĪŖ2CoCl2+2NH4Cl+10NH3+H2O2$\frac{\underline{\;“߻ƼĮ\;}}{\;}$2[Co£ØNH3£©6]Cl3+2H2O£»Ė«ŃõĖ®Ņ×·Ö½ā”¢ĘųĢåµÄČܽā¶ČĖę×ÅĪĀ¶ČµÄÉżø߶ų½µµĶ£¬ĖłŅŌXµÄÖʱø¹ż³ĢÖŠĪĀ¶Č²»Äܹżøߣ¬
¹Ź“š°øĪŖ£ŗ+3£»2CoCl2+2NH4Cl+10NH3+H2O2$\frac{\underline{\;“߻ƼĮ\;}}{\;}$2[Co£ØNH3£©6]Cl3+2H2O£»ĪĀ¶ČŌ½øß¹żŃõ»ÆĒā·Ö½ā”¢°±ĘųŅŻ³ö£¬¶¼»įŌģ³É²āĮæ½į¹ū²»×¼Č·£®
µćĘĄ ±¾Ģāæ¼²éĮĖĪļÖŹŗ¬ĮæµÄ²ā¶Ø£¬Éę¼°ÄŃČÜĪļµÄČܽāĘ½ŗā”¢Ńõ»Æ»¹Ō·“Ó¦”¢ĪļÖŹŗ¬ĮæµÄ²ā¶ØµČÖŖŹ¶µć£¬Ć÷Č·ŹµŃéŌĄķŹĒ½ā±¾Ģā¹Ų¼ü£¬ÖŖµĄÖøŹ¾¼ĮµÄєȔ·½·Ø£¬ĢāÄæÄѶČÖŠµČ£®



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¶ąŃ”Ģā
| A£® | ±źæöĻĀ1molCCl4ĖłÕ¼Ģå»żŌ¼ĪŖ22.4L | |
| B£® | ²»Ķ¬·ÖÉ¢ĻµĘä¶ŌÓ¦·ÖÉ¢ÖŹµÄĪ¢Į£Ö±¾¶²»Ķ¬ | |
| C£® | 1molO2Óė1molO3Ėłŗ¬µÄŌ×ÓŹżÄæĻąµČ | |
| D£® | NaCl¾§ĢåŗĶČŪ»ÆµÄNaCl¶¼ŹōÓŚµē½āÖŹ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | AlŅ×µ¼µē£¬ŹōÓŚµē½āÖŹ | |
| B£® | BaSO4ÄŃČÜÓŚĖ®£¬ŹōÓŚ·Ēµē½āÖŹ | |
| C£® | HClµÄĘųĢåŗĶŅŗĢ嶼²»µ¼µē£¬µ«HClŹōÓŚµē½āÖŹ | |
| D£® | SO2ČÜÓŚĖ®Äܵ¼µē£¬ĖłŅŌSO2ŹĒµē½āÖŹ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | Na+”¢CO32-”¢Cu2+”¢SO42- | B£® | K+”¢Fe3+”¢SO42-”¢OH- | ||
| C£® | Ca2+”¢NO3-”¢Cl-”¢K+ | D£® | Ba2+”¢K+”¢MnO4-”¢NO3- |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | ¢Ū¢Ż | B£® | ¢Ł¢Ū¢Ü | C£® | ¢Ś¢Ū¢Ü | D£® | ¢Ś¢Ū¢Ü¢Ż |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā

 £®
£®²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā


²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | N2”¢H2ĶźČ«×Ŗ»ÆĪŖNH3 | B£® | N2”¢H2”¢NH3µÄÅضČŅ»¶ØĻąµČ | ||
| C£® | Õż”¢Äę·“Ó¦ĖŁĀŹĻąµČĒŅµČÓŚĮć | D£® | N2”¢H2”¢NH3µÄÅØ¶Č²»ŌŁøıä |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | øɱłæÉÉż»Ŗ”¢Ņŗ°±æÉĘū»Æ | |
| B£® | øɱłÓŠĖįŠŌ”¢Ņŗ°±ÓŠ¼īŠŌ | |
| C£® | øɱłÉż»Ŗ”¢Ņŗ°±Ęū»ÆŹ±æɷųö“óĮæČČ | |
| D£® | øɱłÉż»Ŗ”¢Ņŗ°±Ęū»ÆŹ±æÉĪüŹÕ“óĮæČČ |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com