
 |  |  |  |
| A������ | B���ܽ� | C��ת�� | D��ҡ�� |
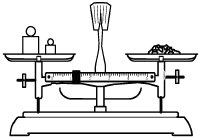
���� ��1������m=CVM������Ҫ���ʵ�������
��2����������һ�����ʵ���Ũ����Һһ�㲽��ѡ����Ҫ��������
��3����������һ�����ʵ���Ũ����Һ��ȷ�IJ���������
��4���������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$���з�����
��� �⣺��1������1000mL 0.2mol/LNaCl��Һ����Ҫ�Ȼ��Ƶ�����m=0.2mol/L��1L��58.5g/mol=11.7g��
�ʴ�Ϊ��11.7��
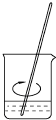
��2������һ�����ʵ���Ũ����Һһ�㲽�裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȣ��õ���������
������ƽ��ҩ�ס���Ͳ���ձ�����������1000 mL����ƿ�� ��ͷ�ιܣ����Ի�ȱ�ٵ�������
1000 mL����ƿ�� ��ͷ�ιܣ�
�ʴ�Ϊ��1000 mL����ƿ�� ��ͷ�ιܣ�
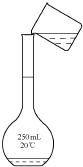
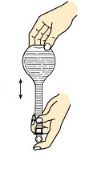
��3������һ�����ʵ���Ũ����Һһ�㲽�裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȣ�
A������ʱӦ��ѭ�������룬��A����
B���ܽ�ʱӦ�ò��������裬��B��ȷ��
C����ҺʱӦ�ò�������������C����
D��ҡ��ʱ��ʳָ��סƿ�����������ҵߵ�����D��ȷ��
�ʴ�Ϊ�����ݣ� BD��
��4����������Һʱ��ϴ�ɾ�������ƿ�Ŀ̶���֮�²�������������ˮ�������ʵ����ʵ�������Һ�����������Ӱ�죬��ҺŨ�Ȳ��䣻
��ҡ�Ⱥ���Һ����ڿ̶��ߣ��ּ�����������ˮ��̶�����ƽ��������Һ���ƫ����ҺŨ��ƫ�ͣ�
�ʴ�Ϊ����Ӱ�죻 ƫ�ͣ�
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���ȷ���ƹ��̼����������ǽ���ؼ�����Ŀ�ѶȲ���ע���������ķ����ͼ��ɣ�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Cԭ�ӵĹ����ʾʽ�� | |
| B�� | Nԭ�ӹ����ʾʽ�� | |
| C�� | Cr��̬ԭ�Ӻ�������Ų�ʽ��[Ar]3d54s1 | |
| D�� | Caԭ�ӵĵ����Ų�ʽ��[Ar]3d2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��ϩ��ʹ���Ը��������Һ��ɫ����������ʹ����ɫ | |
| B�� | ����ˮ�ܵ���Ļ����ﲻһ�������ӻ����� | |
| C�� | ��Ҫ���ȵķ�Ӧ��һ�������ȷ�Ӧ | |
| D�� | ʯ�͵ķ����ú�ĸ����ǻ�ѧ�仯 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �ܱ�������2molNO��1molO2��ַ�Ӧ������ķ�����Ϊ2NA | |
| B�� | һ���¶��£�1 L 0.50 mol•L-1 NH4NO3��Һ�к���ԭ�Ӹ���ΪNA | |
| C�� | ��������ֽ��Ƶ�1.12 L O2��ת�Ƶ�����ĿΪ 0.2 NA | |
| D�� | 235g����${\;}_{92}^{235}$U�����ѱ䷴Ӧ��${\;}_{92}^{235}$U+${\;}_{0}^{1}$n$\stackrel{�ѱ�}{��}$${\;}_{38}^{90}$Sr+${\;}_{54}^{136}$Xe+10${\;}_{0}^{1}$n�������������ӣ�${\;}_{0}^{1}$n����Ϊ10NA |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 �Թ�ҵ��ˮ��������ˮ���д����Ƿ�ֹˮ����Ⱦ������ˮ�ʵ���Ҫ��ʩ֮һ��
�Թ�ҵ��ˮ��������ˮ���д����Ƿ�ֹˮ����Ⱦ������ˮ�ʵ���Ҫ��ʩ֮һ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �٢� | B�� | �ۢ� | C�� | �ۢ� | D�� | �٢ڢ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ͭƬ����FeCl3��Һ�У�Cu+2Fe3+�T2Fe2++Cu2+ | |
| B�� | �������Ca CO3��CO32-+2H+�TH2O+CO2�� | |
| C�� | Na2O2��H2O��Ӧ��Na2O2+H2O�T2Na++2OH-+O2�� | |
| D�� | ��Fe��OH��2�м�������ϡ���Fe��OH��2+2H+�TFe2++2H2O |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com