
����25��ʱ0.1 mol/L�İ�ˮ����ش��������⣺
(1)����ˮ�м�����������粒��壬��ʱ��Һ��![]() ________(�������С�����䡱)��
________(�������С�����䡱)��
(2)����ˮ�м���ϡ���ᣬʹ��ǡ����ȫ�кͣ�д����Ӧ�����ӷ���ʽ________��������Һ��pH________7(���������������)�������ӷ���ʽ��ʾ��ԭ��________��
(3)����ˮ�м���ϡ��������Һ��pH��7����ʱ![]() ����
����![]() ��________��
��________��
(4)����ˮ�м���pH��1�����ᣬ�Ұ�ˮ������������Ϊ1��1����������Һ�и��������ʵ���Ũ���ɴ�С��˳����________��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(1)����ˮ�м�����������粒��壬��ʱ��Һ��![]() ____________(�������С�����䡱)��
____________(�������С�����䡱)��
(2)����ˮ�м���ϡ���ᣬʹ��ǡ����ȫ�кͣ�д����Ӧ�����ӷ���ʽ__________________��������Һ��pH____________7(�������������=��)�������ӷ���ʽ��ʾ��ԭ��______________
_____________________________________________________________________��
(3)����ˮ�м���ϡ��������Һ��pH=7����ʱc(![]() )=a mol��L-1����c(
)=a mol��L-1����c(![]() )=_______��
)=_______��
(4)����ˮ�м���pH=1�����ᣬ�Ұ�ˮ������������Ϊ1��1����������Һ�и��������ʵ���Ũ���ɴ�С��˳����______________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�갲��ʡ���������ѧ�߶���ѧ����������5��ѧ�Ծ����������� ���ͣ������
(14��)����25 ��ʱ0.1 mol/L�İ�ˮ����ش��������⣺
(1)����ˮ�м�����������粒��壬��ʱ��Һ��________(�������С�����䡱)��
(2)����ˮ�м���ϡ���ᣬʹ��ǡ���кͣ�д����Ӧ�����ӷ���ʽ________________ ________________________________________________________��
������Һ��pH________7(�>����<������)�������ӷ���ʽ��ʾ��ԭ��____________ ____________________________________________________________��
(3)����ˮ�м���ϡ��������Һ��pH��7����ʱc(NH4��)��a mol/L����c(SO42��)��________��
(4)����ˮ�м���pH��1�����ᣬ�Ұ�ˮ������������Ϊ1��1����������Һ�и��������ʵ���Ũ���ɴ�С�Ĺ�ϵ��__________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��ɽ��ʡ��̨�и�����ѧ����ĩͳ����ѧ�Ծ��������棩 ���ͣ������
�ɶ����ڳ���Ԫ���γɵĴ�����A��B��C��D��E��F��Xת����ϵ����ͼ��ʾ��ij��������ȥ����

��֪��B��XΪ���ʣ�������DΪ��ɫҺ�壬A��B��ͬһ��Ԫ�ء�
��ش��������⣺
��1����E�����Ǵ�����Ⱦ�F��һԪǿ�ᡣ
��д��E��F��Ӧ�Ļ�ѧ����ʽ�� ��
������25��ʱ0.1 mol��L��1 A��ˮ��Һ���������м�������0.1 mol��L��1��ϡ���ᣬ��������Һ�и����ӵ����ʵ���Ũ���ɴ�С��˳���� ��
���ڳ����£���V1 L pH��a��A��ˮ��Һ�м���V2 L pH��b�����ᣬ��a��b��14����ǡ����ȫ��Ӧ����V1��V2�Ĺ�ϵΪVl V2���������������������������ȷ����������pH��������Һ������Ƚϣ���ˮ�������c(H��)ǰ��Ϊ���ߵ�108������������Һ��pH�� ��
��2����E���岻�Ǵ�����Ⱦ�F�Ƕ�Ԫ���ᡣ
��B����Ԫ�������ڱ��е�λ�� ��
��д����������C��Ӧ�Ļ�ѧ����ʽ�� ������������Eͨ������������Һ�еò�����F��F��Ksp��2.8��10��9���ֽ��ó�������0.1 mol��L��1��CaCl2��Һ�У���Ksp �����������С�����䡱������ʱ����ɲ�����F������������Һ�е�Ũ��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��ӱ�ʡʯ��ׯ�и߶���ѧ�����п������ƻ�ѧ�Ծ��������棩 ���ͣ������
����25 ��ʱ0.1 mol/L�İ�ˮ����ش��������⣺
(1)����ˮ�м�����������粒��壬��ʱ��Һ��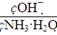 _________(�������С�����䡱)��
_________(�������С�����䡱)��
(2)����ˮ�м���ϡ���ᣬʹ��ǡ����ȫ�кͣ�д����Ӧ�����ӷ���ʽ____________________��������Һ��pH___ 7(���������������)�������ӷ���ʽ��ʾ��ԭ��_________________��
(3)����ˮ�м���ϡ��������Һ��pH��7����ʱc(NH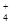 )��a mol/L����c(SO
)��a mol/L����c(SO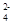 )��__________��
)��__________��
(4)����ˮ�м���pH��1�����ᣬ�Ұ�ˮ������������Ϊ1��1����������Һ�и��������ʵ���Ũ���ɴ�С��˳����___ _______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�츣��ʡ�߶���ѧ����ĩ���Ի�ѧ�Ծ��������棩 ���ͣ������
(14��) ����25 ��ʱ0.1 mol/L�İ�ˮ����ش��������⣺
(1)����ˮ�м�����������粒��壬��ʱ��Һ�� c(OH��) / c(NH3��H2O) ______ (�������С�����䡱)��
(2)����ˮ�м���ϡ���ᣬʹ��ǡ���кͣ�д����Ӧ�����ӷ���ʽ��
_____________________________________________________________________��
������Һ��pH________7(�>����<������)�������ӷ���ʽ��ʾ��ԭ��
_________________ ��
(3)����ˮ�м���ϡ��������Һ��pH��7����ʱc(NH4��)��a mol/L����c(SO42��)��________��
(4)����ˮ�м���pH��1�����ᣬ�Ұ�ˮ������������Ϊ1��1����������Һ�и��������ʵ���Ũ���ɴ�С�Ĺ�ϵ��______________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com